Understanding phase change materials for thermal energy storage

As the world searches for practical ways to decarbonize our activities and mitigate associated climate change, approaches to alternative energy are hampered by the intermittent nature of energy sources, such as solar and wind. One possible solution to help boost reliability and adoption of such renewable energy sources is improved energy storage capabilities.
In the Journal of Applied Physics, researchers from Lawrence Berkeley National Laboratory, Georgia Institute of Technology, and the University of California, Berkeley, describe advances in understanding the fundamental physics of phase change materials used for energy storage.
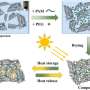
Phase change materials absorb thermal energy as they melt, holding that energy until the material is again solidified. Better understanding the liquid state physics of this type of thermal storage may help accelerate technology development for the energy sector.
"Modeling the physics of gases and solids is easier than liquids," said co-author Ravi Prasher. "Gases are free moving, and solids merely vibrate, but liquids behave more like a solid when melting and more like a vapor as they heat up."
This behavior makes it difficult to model and predict storage-system behavior during the phase change critical to its function.
To best capitalize on phase change phenomena of materials for thermal storage, material parameters, including molecular motion and entropy, must be mathematically described, so behavior and theoretical limits can be predicted. The researchers describe a step toward this predictive power by discussing past literature and new developments in the field of liquid state physics.
"The amount of energy that gets stored during phase change depends on the entropy of melting," said Prasher. "Once you know how to predict the entropy change, you know how to design materials that will cater to specific needs."
Developing high-performance thermal energy storage material is important, as heat energy dominates energy use in buildings and manufacturing. Thermal storage is also safer than many other forms of energy storage, since it does not have the capability to release stored energy rapidly and destructively in the case of a malfunction.
Finally, thermal storage holds promise for functioning at large scales and over long durations, and individualized and/or novel materials can be manufactured to suite specific needs. More sophisticated models are needed to further aid in the ability to screen and create materials for optimal thermal energy storage applications.
More information: Drew Lilley et al, Phase change materials for thermal energy storage: A perspective on linking phonon physics to performance, Journal of Applied Physics (2021). DOI: 10.1063/5.0069342
Journal information: Journal of Applied Physics
Provided by American Institute of Physics