Solvent effect on liquid-phase synthesis of lithium solid electrolytes
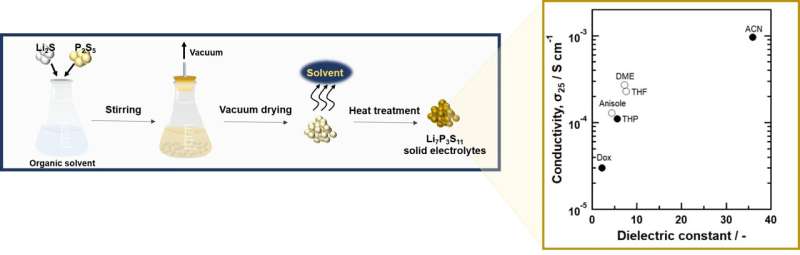
A research group of Department of Electrical and Electronic Information Engineering of Toyohashi University of Technology, consisting of Hirotada Gamo, doctoral course student; Atsushi Nagai, project associate professor; and Atsunori Matsuda, professor, demonstrated that solvent polarity characterized by its dielectric constant plays an important role in the formation of crystalline Li7P3S11 in the liquid-phase synthesis, a solid electrolyte for all-solid-state lithium-ion secondary batteries. Among the solvents systematically studied, the research team found that acetonitrile (ACN) with a high dielectric constant is the best solvent for the liquid-phase synthesis of Li7P3S11, since it enhances the reactivity of lithium thiophosphate and enables the formation of crystalline Li7P3S11 with high conductivity. This achievement is expected to lead to the development of mass production technology for all-solid-state batteries.
Details
All-solid-state batteries are attracting attention as next-generation batteries due to their high safety and energy density and, in particular, they are expected to find application with electric vehicles. The practical application of all-solid-state batteries requires mass production technology for sulfide solid electrolytes with high ionic conductivity. Among the synthesis methods for solid electrolytes, liquid-phase synthesis is the most promising due to its low cost and suitability for mass production. While Li7P3S11 is one of the candidates for solid electrolytes for all-solid-state batteries because of its high ionic conductivity, the effect of solvent on the liquid-phase synthesis of crystalline Li7P3S11 has not been systematically investigated.
Therefore, the research group selected eight organic solvents with various phisico-chemical properties and revealed the effect of the solvent species on the reaction in the liquid-phase synthesis of Li7P3S11. Among the solvents studied, they found that the use of 1,4-dioxane (Dox), tetrahydropyran (THP) and ACN as solvents results in the formation of crystalline Li7P3S11 and a higher conductivity tends to be obtained in organic solvents with higher dielectric constants. The higher the dielectric constant of the solvent applied to the sample, the higher the temperature at which desolvation occurred during the heat treatment process. This indicates that the dielectric constant of the solvent is an indicator of the strength of the chemical interaction with lithium thiophosphate. In general, reactivity in solvents is characterized by dielectric constant, dipole moment, donor number, etc. In the liquid-phase synthesis of Li7P3S11, it was found that high reactivity is observed in solvents with a high dielectric constant.
From the viewpoint of the drying process, a solvent with a low boiling point is preferable. Based on these combined properties, the research group demonstrated that ACN is the best reaction solvent for the liquid-phase synthesis of crystalline Li7P3S11 with high conductivity.
Future Outlook
In the liquid-phase synthesis of Li7P3S11, which is a promising candidate of solid electrolyte for all-solid-state batteries, the research team believes that it has found an important basis for solvent selection to achieve high conductivity in Li7P3S11 solid electrolytes. In accordance with the basis for solvent selection found from the study, the research team intends to utilize it for the development of mass production technology for all-solid-state batteries.
The research was published in Scientific Reports
More information: Hirotada Gamo et al, The effect of solvent on reactivity of the Li2S–P2S5 system in liquid-phase synthesis of Li7P3S11 solid electrolyte, Scientific Reports (2021). DOI: 10.1038/s41598-021-00662-3
Journal information: Scientific Reports
Provided by Toyohashi University of Technology




















