Molecular interfaces as building blocks for innovative sensors and data storage devices
Molecular interfaces formed between metals and molecular compounds have enormous potential as building blocks for future opto-electronics and spin-electronics devices. Transition metal phthalocyanine and porphyrin complexes are promising components for such interfaces. Scientists at Forschungszentrum Jülich, together with a team of international scientists, have been working to develop a model system for designing such devices with unique functions and enhanced performance by stabilizing and controlling the spin and oxidation states in the complexes with nanoscale precision. Among other things, they discovered a mechanism that can be used in the future to store information in porphyrins or to develop extremely sensitive sensors to detect toxic nitrogen dioxide.
Some of the most important processes in biological systems are catalyzed by enzymes containing metal ions, where unexpected reactivity corresponds to low oxidation states. For example, porphyrins, a class of dye molecules, are involved in photosynthesis in plants and oxygen transport in red blood cells. Inspired by their biological functions, scientists have allocated porphyrins a wide range of technological uses. However, any practical application of these organometallic complexes in the sphere of technology demands nanometric-scale control of the molecular properties to be exploited.
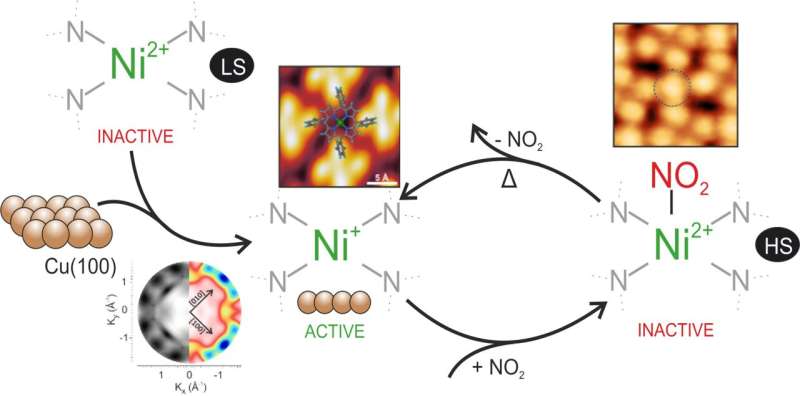
A group of scientists from Forschungszentrum Jülich has been working on these systems for some time with the goal of fine-tuning their electronic and magnetic properties and understanding the mechanisms that govern the interactions at the interface. "We have taken the first step in this direction by coupling nickel-porphyrin with copper, which is a highly interactive surface. This unique combination results in some really interesting properties: For example, copper promotes a significant charge transfer in porphyrin. Furthermore, it triggers the reduction of the central metal, nickel, bringing the characteristics of this system closer to the biological systems that inspired us in the first place. As a result, we wondered, why not go even further, making use of Ni(I)'s high reactivity?" explains Dr. Vitaliy Feyer from Jülich's Peter Grünberg Institute.
Indeed, the unsaturated low valence Ni(I) metal ions at this interface are available for catalysis, and the attachment of axial ligands, such as small diatomic molecules, provides the possibility for further controlling the oxidation and spin states. What seemed to be a simple approach has resulted in intriguing discoveries: For example, exposing the molecular interface to a low dosage of nitrogen dioxide resulted in the nickel ion switching to a higher spin state. Even in a buried multilayer system, the chemically active low valence nickel ion can be functionalized with nitrogen dioxide, providing selective tuning of the metal center's electronic properties.
The axial ligand coordination spin switching at the interface is a reversible process, and the pristine state can be restored by the mild annealing of the interface. While nickel works as a reversible spin switch at room temperature, the electronic structure of the macrocycle backbone, where the frontier orbitals are primarily localized, is unaltered. "The reason for this is that the porphyrin's strong contact with the substrate appears to behave as an energy counterpart, preventing further geometric modifications caused by the so-called surface trans-effect," says Iulia Cojocariu, Ph.D. student at the Peter Grünberg Institute. This method has never been observed at room temperature before and has the potential to be exploited in the future to store information in porphyrins or to construct extraordinarily sensitive sensors for detecting hazardous substances such as nitrogen dioxide.
The research was published in Small.
More information: Henning Maximilian Sturmeit et al, Room‐Temperature On‐Spin‐Switching and Tuning in a Porphyrin‐Based Multifunctional Interface, Small (2021). DOI: 10.1002/smll.202104779
Journal information: Small
Provided by Forschungszentrum Juelich