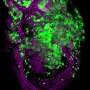
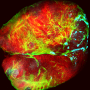
Relationship between chromosomal instability and senescence revealed in Drosophila
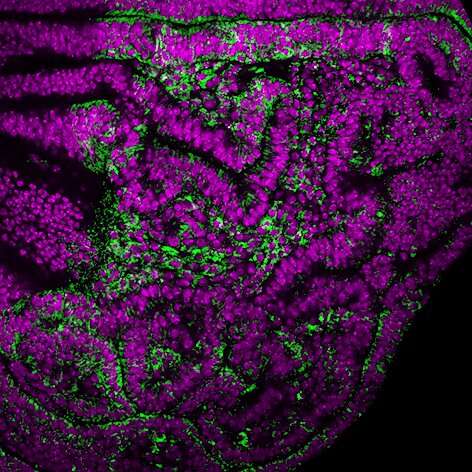
Chromosomal instability is a feature of solid tumors such as carcinoma. Likewise, cellular senescence is a process that is highly related to cellular aging and its link to cancer is becoming increasingly clear. Scientists led by ICREA researcher Dr. Marco Milán at IRB Barcelona have revealed the link between chromosomal instability and cellular senescence.
"Chromosomal instability and senescence are two characteristics common to most tumors, and yet it was not known how one related to the other. Our studies indicate that senescence may be one of the intermediate links between chromosomal alterations and cancer," says Dr. Milan, head of the Development and Growth Control laboratory at IRB Barcelona.
"The behavior we saw in cells with chromosomal instability made us think that they could be senescent cells, and indeed that was the case," says Dr. Jery Joy, first author of the article published in Developmental Cell.
The study has been conducted on the fly Drosophila, an animal model commonly used in biomedicine, and the mechanisms described may help to understand the contribution of chromosomal instability and senescence to cancer, and facilitate the identification of possible therapeutic targets.
Reversing the effects of chromosomal instability
The researchers from the Development and Growth Control lab have shown that in an epithelial tissue with high levels of chromosomal instability, those cells with an altered balance of chromosome number detach from their neighboring cells and enter senescence. Senescent cells are characterized by a permanently halted cell cycle and by the secretion of a large number of proteins. This abnormal secretion of proteins alters the surrounding tissue, alerting the immune system and causing inflammation.
If the senescent cells are not eliminated by the body immediately, they promote abnormal growth of the surrounding tissue, leading to malignant tumors. "If we identify the mechanisms by which we can lower the number of senescent cells, then we will be able to reduce the growth of these tumors," says Dr. Milan. "In fact, this study shows that this is possible, at least in Drosophila," says Dr. Joy.
Cells with an unbalanced number of chromosomes accumulate a high number of aberrant mitochondria, and therefore, a high level of oxidative stress, which in turn activates the JNK signaling pathway, triggering entry into senescence. "We have shown that reducing this high anomalous number of mitochondria, or regulating the oxidative stress that they induce, is sufficient to decrease the number of senescent cells and the negative effects of chromosomal instability," reiterates Dr. Joy.
These findings open new avenues of research to find therapeutic targets and reduce senescence levels caused by chromosomal instability in solid tumors.
Extrapolating from the fly to mammals
The vinegar fly, Drosophila melanogaster, is a widely used in biomedicine. It is a valuable animal model in cancer research because of its short life cycle, the availability of a large number of genetic tools, and the presence of the same genes as in humans, but with a lower level of redundancy.
In fact, experiments designed to dissect the causal relationship between cellular behavior or characteristics in human tumors, such as chromosomal instability and senescence, are more easily analyzed in this model organism.
Future laboratory work will continue to dissect the molecular mechanisms responsible for cellular behaviors found in solid tumors of epithelial origin produced by the mere induction of chromosomal instability. "The more we understand the biology of a tissue subjected to chromosomal instability and the molecular mechanisms responsible for the cellular behaviors that emerge and give rise to malignant tumors, the greater our chances of designing effective therapies and reducing the growth and malignancy of human carcinomas," concludes Dr. Milan.
More information: Jery Joy et al, Proteostasis failure and mitochondrial dysfunction leads to aneuploidy-induced senescence, Developmental Cell (2021). DOI: 10.1016/j.devcel.2021.06.009
Journal information: Developmental Cell