Evolution: Fruit flies find two routes to the same destination
Fruit flies have found at least two solutions to the problem of sorting their sex chromosomes: a matter of life and death.
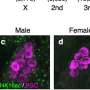
Sex determination in animals often depends on the unequal segregation of specific chromosomes. Female cells generally possess two X chromosomes, while male cells contain one X and one Y chromosome. The latter, which is inherited from the male parent, has far fewer genes than the X. In the fruit fly Drosophila, male cells make up for the fact that they have only one X chromosome by boosting the level of expression of all of its genes by a factor of 2. This phenomenon, which is known as dosage compensation, requires that the X chromosome in males be regulated differently from all the others. A team of molecular biologists at Ludwig-Maximilians-Universitaet (LMU) in Munichs Biomedical Center led by Professor Peter Becker has now shown that, over the course of 40 million years, members of the genus Drosophila have discovered at least two different ways of making this vital distinction.
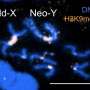
"In light of the significance of dosage compensation, one might expect that the principles behind the specific recognition of the X chromosome in males would be highly conserved," says Becker. "In other words, the process should work in essentially the same way in all Drosophila species. However, when we compared the two species Drosophila melanogaster and Drosophila virilis, we discovered, to our surprise, that they use distinct mechanisms for this purpose." Significantly, the primary components involved in dosage compensation—the proteins MSL2 and CLAMP, together with the non-coding RNA roX—are found in both species. So their last common ancestor presumably possessed the genes that code for these products.
The two species diverged about 40 million years ago, and since then they have evolved in parallel. The new study shows that, during this period, the mediators of dosage compensation and their binding sites on the X chromosome have evolved in different ways. As a result, the relative influence of, and the interactions between, the components have changed. Among other things, in D. melanogaster the copy numbers of certain DNA sequences on the X chromosome have increased. In parallel, the DNA-binding domain of the MSL2 protein has acquired the ability to recognize these sequences, and they now play a critical role in the recognition of the X chromosome in this species.
In D. virilis, on the other hand, these sequences have not been amplified. Their recognition by MSL2 therefore depends on its interaction with the CLAMP protein to a much greater extent than in the case of D. melanogaster—even though the CLAMP protein can also bind to many sequences on the other chromosomes. "We assume that the non-coding roX RNA inhibits the binding of MSL2 at these sites," says Becker. The study has therefore uncovered a new role for this RNA. Up until now, researchers had assumed that roX comes into play not at the level of sequence recognition, but at a later stage in the dosage compensation process.
These findings have interesting evolutionary implications. "As the sex chromosomes continue to diversify, the emergence of alternative but equally effective solutions to the problem of balancing the activity of the genome demonstrates that evolution is not a deterministic process," Becker says.
More information: Raffaella Villa et al, Divergent evolution toward sex chromosome-specific gene regulation in Drosophila, Genes & Development (2021). DOI: 10.1101/gad.348411.121
Journal information: Genes & Development
Provided by Ludwig Maximilian University of Munich