A new culprit in antibacterial resistance: Cysteine persulfide
A joint research project based in Kumamoto University, Japan has developed a new, highly sensitive analytical method that can detect degraded β-lactam antibacterial agents used in the treatment of bacterial infections. With this method, researchers found that reactive sulfur species produced by bacteria degrade and inactivate β-lactam antibiotics.
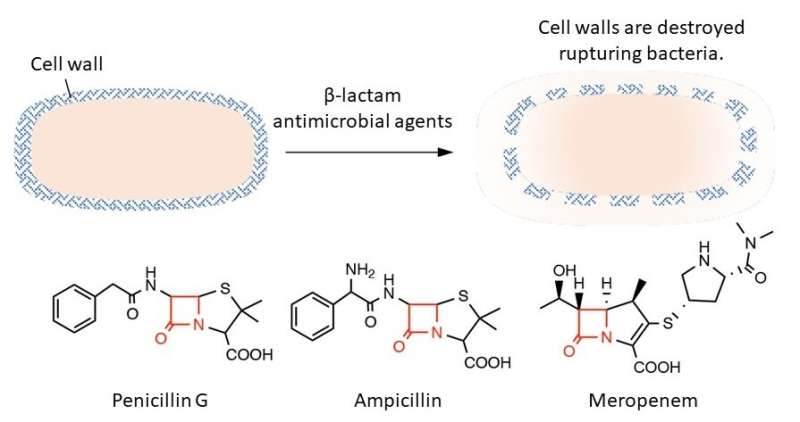
Bacteria are different from animal cells in that their outer layer is covered with a rigid structure called a cell wall. β-lactam antimicrobial agents interfere with the processes that form the cell wall. This results in bacteria no longer being able to withstand their own internal pressure so they rupture and die. β-lactam antimicrobial agents are very potent because they selectively inhibit bacterial cell wall synthesis and have few side effects on hosts such as humans. These antimicrobial agents have a common structure called the β-lactam ring that is essential for inhibiting cell wall development. If this ring is degraded, the antimicrobial effect disappears.
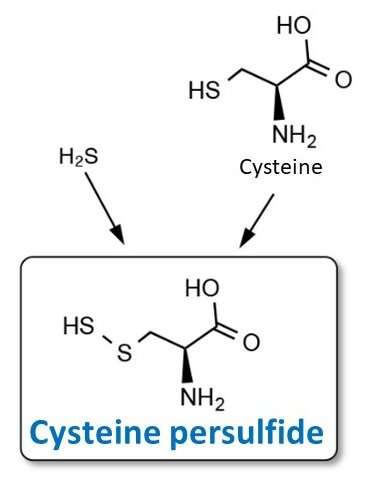
Previous studies have reported that hydrogen sulfide (H2S), which bacteria produce during sulfur metabolism, reduces their susceptibility to antimicrobial agents leading to resistance. However, the detailed mechanism causing this are not yet understood. Researchers at Kumamoto University previously showed that the molecule cysteine persulfide, a combination of H2S and the amino acid cysteine, has an extremely potent antioxidant effect that is not found in H2S or cysteine alone.
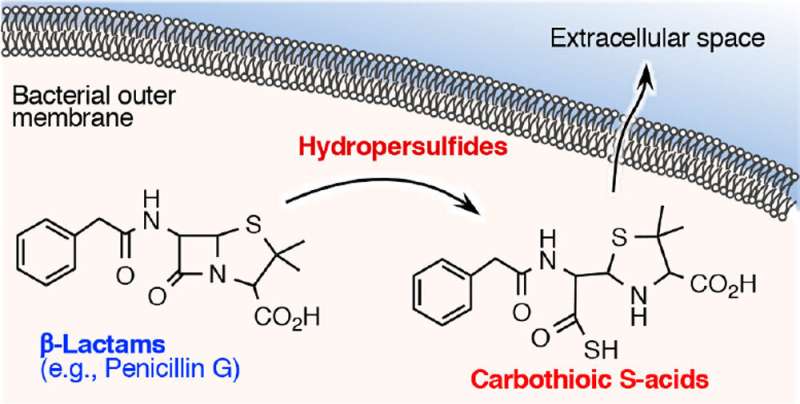
In this study, researchers examined how this reactive sulfur species is involved in the acquisition of resistance to β-lactam antibiotics. They discovered that β-lactam antibiotics such as penicillin G, ampicillin, and meropenem (carbapenem antibiotics) rapidly lose bactericidal activity when exposed to cysteine persulfide but not with hydrogen sulfide. A detailed study of the reaction between β-lactam antimicrobial agents and cysteine persulfide revealed that the β-lactam ring, which is essential for bactericidal action, decomposes and a sulfur atom is inserted into part of the ring creating carbothioic acid. The production of carbothioic acid from a β-lactam antimicrobial agent appears to be a novel degradation metabolite.
Researchers thus developed a highly sensitive analytical method to detect and quantify carbothioic acid using mass spectrometry, and then analyzed carbothioic acid production from bacteria that were exposed to β-lactam antimicrobials. They found that bacteria can absorb antimicrobial agents and use cysteine persulfide to degrade the agents into carbothioic acid which is then discharged. This is believed to be a previously undescribed inactivation and degradation mechanism of β-lactam antimicrobial agents into carbothioic acid by cysteine persulfide.
"Our newly developed analytical method makes it possible to quantify the amount of carbothioic acid discharged from bacteria with high sensitivity," said Professor Tomohiro Sawa, who led the study. "We believe it will be possible to screen for compounds that inhibit bacterial synthesis of cysteine persulfide by using carbothioic acid as a biomarker. Such a cysteine persulfide synthesis inhibitor in combination with β-lactam antibiotics is expected to inhibit antibiotic degradation and result in successful treatments with a lower concentration of β-lactam antibiotics. This should also help to reduce the emergence of new resistant bacteria."
This research was posted online in ACS Chemical Biology on 30 March 2021.
More information: Katsuhiko Ono et al, Cysteine Hydropersulfide Inactivates β-Lactam Antibiotics with Formation of Ring-Opened Carbothioic S-Acids in Bacteria, ACS Chemical Biology (2021). DOI: 10.1021/acschembio.1c00027
Journal information: ACS Chemical Biology
Provided by Kumamoto University