Single cell sequencing unravels molecular identity of rare renal cell types
Jaakko Patrakka and his research group at the Department of Laboratory Medicine have together with Professor Rickard Sandberg's group at the Department of Cell and Molecular Biology got a publication accepted in Nature Communications about single cell sequencing unravels molecular identity of rare renal cell types.
Kidney tissue is very heterogenous with >30 different distinct cell types. This has made it difficult to define molecular fingerprints of individual renal cell types as expression levels obtained through analysis of whole kidney tissue generates data averaged across large populations of cells. To overcome this challenge, we have developed single cell RNA-sequencing protocols in mouse and human kidney tissue, with a special focus on the ultrafiltration unit of the kidney, the glomerulus.
What are the most important results in the published study?
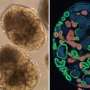
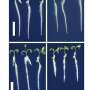
We detected previously unknown heterogeneity in glomerular mural cells (mesangial cells). This is very interesting as mesangial cell injury is known to play a key role in the development of most chronic kidney diseases. Our study re-writes the definition of the mesangial cell and significantly renew our knowledge on this unique cell type. Another major finding in our study was that human kidney cells had very different molecular signatures from their corresponding mouse cells. For instance, many disease-associated human cell-specific genes were not expressed by same cell types in mouse. These differences can explain many translational challenges in the field and our data can hopefully guide the choice of animal models in future studies.
What is your/your group's and KI:s part in the study?
Our group had an overall responsibility for running the project. We optimized and performed single cell capture, performed downstream analyses, validation experiments and functional studies. Bioinformatic analyses were performed together with professor Rickard Sandberg´s group at the Department of Cell and Molecular Biology. Human renal biopsy material was collected at the Division of Pathology at Karolinska University Hospital. High resolution imaging of renal tissue was done in collaboration with Scilifelab in Solna and University of Cologne in Germany.
What happens now, what is the next step?
Obviously, single cell transcriptomics can be applied to study many different biological and clinical questions in nephrology. One outstanding question is about reno-protective drugs: why do they preserve kidney function? Currently used drugs are based largely on repurposing of existing therapeutics and are neither kidney target–based nor designed to address the underlying molecular mechanisms. Single cell profiling of kidneys treated with these drugs can unravel which cell type(s) and mechanisms are mediating their reno-protective effects. This can open up possibilities to develop novel, more targeted kidney therapeutics.
We also want to apply single cell profiling to study regeneration in kidney tissue. This is a highly controversial topic as we still do not know whether some major renal cell types can be regenerated or not. I believe that single cell profiling of potential progenitor populations can give answers to some important pending questions in this field.
More information: Bing He et al. Single-cell RNA sequencing reveals the mesangial identity and species diversity of glomerular cell transcriptomes, Nature Communications (2021). DOI: 10.1038/s41467-021-22331-9
Journal information: Nature Communications
Provided by Karolinska Institutet