Stuck stem cells are no good at making blood
The magic of iPS cells is that they can be expanded into large numbers and differentiated into all cell types, so that an initially small number of iPS cells can be used to produce a massive number of cells. However, some cells have proven easier to make than others, which has significant implications on the cost of translating iPS cell research to clinical therapies. A new study by the Koji Eto lab reports that the choice of laminin, a ubiquitous molecule in the human body and commonly used in iPS cell therapies, influences the hematopoietic differentiation of the iPS cells.
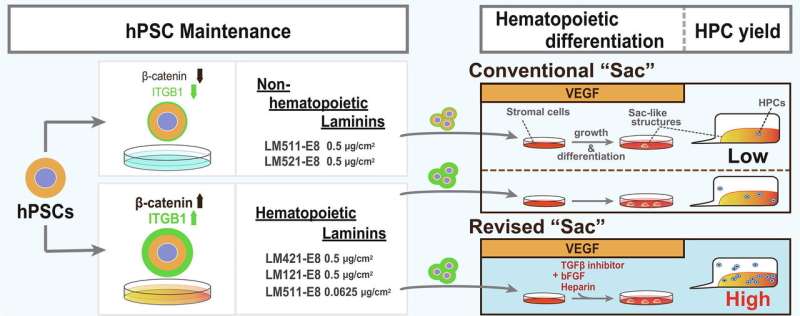
Before differentiating iPS cells, the environment in which the cells are maintained, or cultured, is critical. For research purposes, scientists culture iPS cells on mouse embryonic fibroblasts. These "feeder" cells provide nutrients to the iPS cells. However, for clinical purposes, the iPS cells must be maintained in feeder-free conditions. Scientists have discovered a suitable substitute for mouse embryonic fibroblasts, which is laminin-511 E8 protein fragments, or LM511-E8.
Professor Koji Eto is leading a major project at CiRA that uses LM511-E8 to differentiate several blood cell types from iPS cells, including the platelets that his team has transfused into a patient. The basis of this differentiation is the Sac method, which Eto discovered more than 10 years ago.
"The Sac method is for the hematopoietic differentiation of iPS cells. It follows the primitive streak, mesoderm, and hemogenic endothelium states to produce hematopoietic progenitor stem cells," he explained.
In other words, the Sac method attempts to recapitulate natural hematopoiesis to produce hematopoietic progenitor cells, a cell type from which all other blood cells can be derived.
The method is not without flaws, however.
"We noticed the number of hematopoietic progenitor cells we obtain is small compared to the number of iPS cells," Eto added.
As cells differentiate, their propensity to proliferate reduces. iPS cells have the highest proliferation rate, and progenitor cells have good proliferation ability too. Platelets, on the other hand, do not proliferate. Therefore, a low hematopoietic progenitor cell yield will increase the time and cost of collecting the more than one hundred billion platelets needed for a transfusion.
LM511-E8 is just one of many types of laminins, although it is the only one approved for iPS cell clinical application to date. Laminins are adhesion proteins found throughout the body. They consist of α, β, and γ chains, and each chain is broken further into subsets; LM511-E8 marks α5 chain, β1 chain, and γ1 chain.
In stem cell research, laminins can be thought of as flooring. For the family with a baby beginning to roll and crawl, carpet may be best. On the other hand, the same family who has a puppy too impatient to clean its feet before frolicking inside may prefer tiles. Similarly, the type of laminin should be selected based on the type of cell you want to derive from the iPS cells.
To determine if LM511-E8 was the reason for the low hematopoietic progenitor stem yield, Eto's team investigated nine different types of laminin. They quickly found two, LM421-E8 and LM121-E8, could improve the yield.
Interestingly, LM421-E8 and LM121-E8 distinguished themselves from the other seven by showing weaker adhesion to the iPS cells. Because neither is currently available for clinical application, the researchers modified the adhesion of LM511-E8, finding that reducing the concentration to one-eighth that used in the original Sac method achieved a yield comparable with LM421-E8 and LM121-E8.
"Serially diluting LM511-E8 caused the cells to express more ITGB1. Activating this receptor enhances canonical Wnt signaling and provides a mechanism for the hematopoietic differentiation of iPS cells," said Dr. Akinori Yuzuhira, the first author of the study.
Wnt signaling is essential for cell growth and migration, especially in the embryo. The study went on to show that chemically modifying the Sac method to enhance Wnt signaling increased the yield of hematopoietic progenitor cells even further.
"iPS cells can be used to make all cell types, but the efficiency varies across countries and labs. One of the reasons can be something simple, like the materials used. Our findings show that hematopoietic differentiation depends on optimal adhesive avidity," said Eto.
More information: Akinori Yuzuriha et al. Extracellular laminin regulates hematopoietic potential of pluripotent stem cells through integrin β1-ILK-β-catenin-JUN axis, Stem Cell Research (2021). DOI: 10.1016/j.scr.2021.102287
Journal information: Stem Cell Research
Provided by Kyoto University