Slc20a1b is essential for hematopoietic stem/progenitor cell expansion in zebrafish
Hematopoietic stem/progenitor cells (HSPCs) include hematopoietic stem cells and several lineage-biased hematopoietic progenitor cells, which can provide all blood cell types in an adult organism. Among them, hematopoietic stem cells have the ability of self-renewal and multi-lineage differentiation, and can rapidly respond under acute hematopoietic conditions. Meanwhile, the hematopoietic progenitor cells can maintain the supply of blood cells in homeostatic hematopoiesis. In a word, HSPCs are the core of the blood system, once their homeostasis is destroyed, it will lead to serious blood diseases and even death. Therefore, the researches associated with the HSPCs can provide a strong support for relevant theoretical studies and clinical treatments.
In order to study the development of the hematopoietic system, researchers often utilize zebrafish as a model, which has the advantages of short growth cycle, large amount of transparent eggs, in vitro fertilization of embryos, easy access to gene editing, and so on. In addition, the hematopoietic system of zebrafish is highly conserved to that of human, which is also the basis for the application of zebrafish in hematopoietic researches.
Back in 2012, Wenqing Zhang's research group and Yiyue Zhang's research group collaboratively carried out forward genetic screening of zebrafish, inducing random gene mutations in zebrafish through compound ENU, and obtaining a series of zebrafish mutants with hematopoietic defects. Based on this research, the cooperation team continues to explore these mutants, digging deeper into which genes are mutated and through which mechanisms these genes cause defects in the hematopoietic system.
Recently, a new research paper of Yiyue Zhang's lab and Wenqing Zhang's lab, named as "Slc20a1b is essential for hematopoietic stem/progenitor cell expansion", was published in Science China Life Sciences. This study revealed that a point mutation of zebrafish slc20a1b gene can significantly reduce the proliferation of HSPCs, resulting in a severe defect of hematopoietic development.
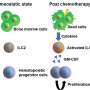
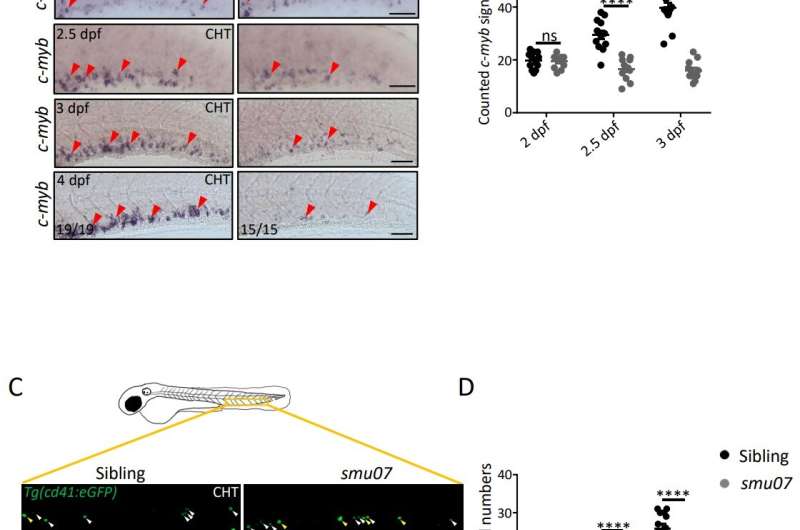
In this research, the researchers first conducted a more in-depth phenotypic identification of the smu07 mutant, obtained by the previous ENU mutagenesis. The hematopoietic defects of the erythroid, myeloid, and lymphoid lineages of the mutant were all present, suggesting that the upstream HSPCs may have been defected. Further investigation showed that the mutant's HSPCs could be produced normally at the initial stages, but then failed to expand continuously, and the number of HSPCs was significantly lower than that of normal zebrafish.
Next, the researchers performed genetic mapping of the mutation site and identified the causative gene to be slc20a1b, in which a single DNA base mutation caused a single amino acid mutation in its protein (D48E).With the help of CRISPR-Cas9 technology, they constructed an additional slc20a1b mutant, which phenotype was identical with the smu07 mutant, verifying that the gene causing the mutation was indeed slc20a1b.
Since slc20a1b is a widely expressed gene, the researchers further explored whether genetic mutations in HSPCs were responsible for its phenotype, or whether other cells in its niche played a dominant role. They found that the mutant HSPCs were unable to expand in a normal environment, while the wild-type HSPCs were able to expand in the mutant environment. This result proved that the defect was caused by the autonomous mutation of the slc20a1b gene in HSPCs.
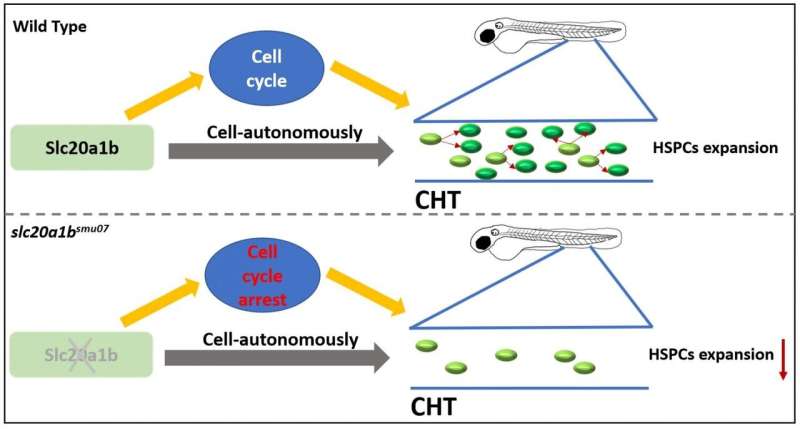
Finally, the researchers investigated the cellular mechanism of the HSPC defects and found that the cell death of mutant HSPCs remained unchanged, but the defect of its proliferation ability prevented the HSPCs from being expanded. The cell cycle of mutant HSPCs was blocked in the G2/M phase and thus could not complete the normal proliferation cycles.
In summary, the authors found that slc20a1b mutant HSPCs autonomously developed an expansion defect, which is featured by cell cycle arrest leading to decreased proliferation (Figure 2), resulting in a severe deficiency of HSPCs and all hematopoietic lineages.
This study is the first to report the indispensable role of slc20a1b gene in HSPCs, revealing a novel regulation gene of the cell cycle of HSPCs.
More information: Jiakui Chen et al, Slc20a1b is essential for hematopoietic stem/progenitor cell expansion in zebrafish, Science China Life Sciences (2021). DOI: 10.1007/s11427-020-1878-8
Journal information: Science China Life Sciences
Provided by Science China Press