Key factor identified that makes worms feel full after a good meal
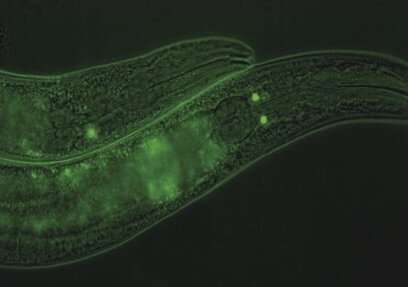
In nematode worms, a key controller allows the worm to sense when it needs food and when it feels full, and then changes its behavior accordingly. Jennifer Tullet of the University of Kent and colleagues report these new findings in a paper published March 4th in PLOS Genetics. They propose that a similar factor may control feelings of fullness in humans.
Deciding when and how much to eat is crucial for maintaining health and to preventing overeating. Our bodies take in complex molecular signals from our nervous, physiological and metabolic systems, which tell us when we're hungry and when to stop eating, but how these signals work are not yet well understood. Tullet and her colleagues used the nematode worm C. elegans, to investigate how the worm's nervous system senses its food status and communicates fullness to the rest of the animal. They identified a new master controller of this system, SKN-1B, which appears to be deeply involved in food-detection and food-related behaviors. SKN-1B is a transcription factor, meaning that it can regulate when other genes are turned on or off. The researchers discovered that it functions by changing hormonal signaling in the worm and activating the network of mitochondria that provides power in each cell. Based on the worm's nutritional needs, SKN-1B can tell the animal to switch between behaviors, such as searching for food, eating and taking a post-meal nap.
The new study suggests the possibility that a similar transcription factor in humans regulates food-sensing and the feeling of being full. Instead of SKN-1B, mammals have NF-E2 related transcription factors, or Nrfs, which scientists think function in metabolism and the process of converting food nutrients into energy. Nrfs also play a role in the phenomenon where animals live longer when they restrict their calories. If future research confirms the role of Nrfs in signaling fullness, then Nrfs may be a new target for developing drugs that control overeating.
The authors add, "We are really excited about this work, understanding the neuroendocrinology of eating and sleeping is so important to lifelong health and wellbeing".
More information: Tataridas-Pallas N, Thompson MA, Howard A, Brown I, Ezcurra M, Wu Z, et al. (2021) Neuronal SKN-1B modulates nutritional signalling pathways and mitochondrial networks to control satiety. PLoS Genet 17(3): e1009358. doi.org/10.1371/journal.pgen.1009358
Journal information: PLoS Genetics
Provided by Public Library of Science