Centimeter-scale porous single-crystalline monoliths with high-density Lewis acid sites to enhance propane dehydrogena
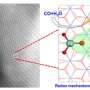
Surface/interface structure and catalytic mechanism are of great significance in many practical catalytic reactions.
Porous single crystals which combine an ordered lattice structure and disordered interconnected pore provide an alternative to create twisted surfaces with clear and high-density active sites in porous microstructures. They present the advantages of clear lattice structure, precise chemical composition and clear termination surface, which demonstrates a huge potential to build continuously twisted and high density active surface structure.
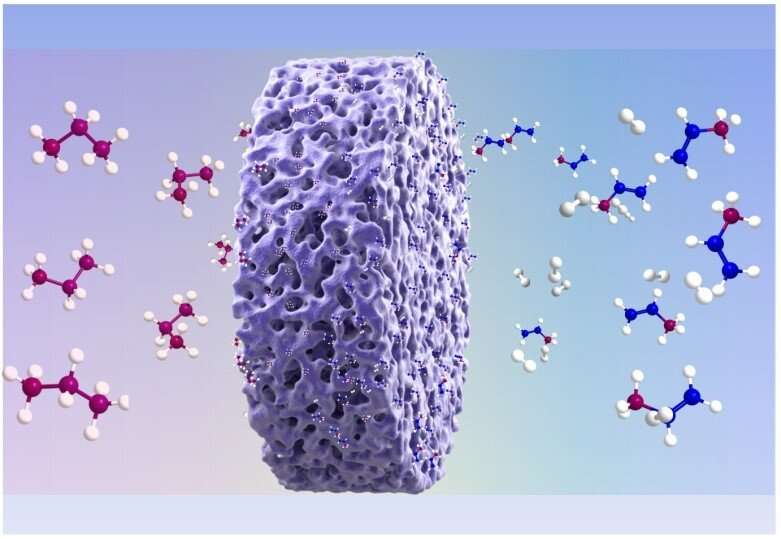
In a study published in Angewandte Chemie International Edition, the research group led by Prof. Xie Kui from Fujian Institute of Research on the Structure of Matter of the Chinese Academy of Sciences grew porous single-crystalline Mo2N and MoN monoliths at 2 cm scale to enhance non-oxidative propane dehydrogenation to propylene.
The creation of high-density Lewis acid sites at the electron-deficient surface effectively improves the catalytic activity at reduced temperatures through control of the unsaturated Mo-N1/6 and Mo-N1/3 coordination structures.
The researchers firstly grew the parent single crystals of MoO3, then removed the periodic target atomic layer O in ammonia atmosphere at 650-700 degrees C, and simultaneously nitrified and reconstructed the framework structure of metastable Mo-O single crystal into the mesoporous Mo2N single crystal.
In the same way, they grew porous MoN single crystals by controlling the temperature, flow rate and pressure of ammonia gas.
Moreover, the researchers visualized the fine structure of the twisted surfaces and the metal nitrogen unsaturated coordination active structure of the porous single crystal.
They found that the top-layer Mo ion with unsaturated Mo-N1/6 and Mo-N1/3 coordination structures create high-density Lewis acid sites at surface, leading to effective activation of C-H bond without over cracking of C-C bond during non-oxidative propane dehydrogenation.
~11% of propane conversion and ~95% of propylene selectivity were demonstrated with porous single-crystalline Mo2N and MoN monoliths at 500 degrees C without degradation being observed even after the operation of 20 hours.
This study provides important reference for the research on the surface structure and catalytic mechanism in actual catalytic reactions.
More information: Kui Xie et al. High‐density Lewis acid sites in porous single‐crystalline monoliths to enhance propane dehydrogenation at reduced temperatures, Angewandte Chemie International Edition (2021). DOI: 10.1002/anie.202100244
Journal information: Angewandte Chemie International Edition
Provided by Chinese Academy of Sciences