Solvation-driven electrochemical actuation
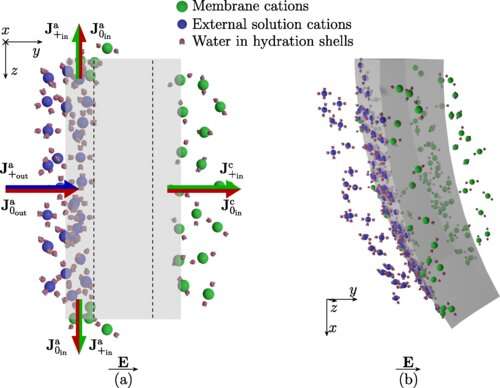
In a new study led by Institute Professor Maurizio Porfiri at NYU Tandon, researchers showed a novel principle of actuation—to transform electrical energy into motion. This actuation mechanism is based on solvation, the interaction between solute and solvent molecules in a solution. This phenomenon is particular important in water, as its molecules are polar: oxygen attracts electrons more than hydrogen, such that oxygen has a slightly negative charge and hydrogen a slightly positive one. Thus, water molecules are attracted by charged ions in solution, forming shells around them. This microscopic phenomenon plays a critical role in the properties of solutions and in essential biological processes such as protein folding, but prior to this study there was no evidence of potential macroscopic mechanical consequences of solvation.
The group of researchers, which also included Alain Boldini, a Ph.D. candidate in the Department of Mechanical and Aerospace Engineering at NYU Tandon, and Dr. Youngsu Cha of the Korea Institute of Science and Technology, proposed that solvation could be exploited to produce macroscopic deformations in materials. To this end, Porfiri and his group utilized ionomer membranes, unique polymeric materials in which negative charges cannot move. Positive ions can easily enter these membranes, while negative ions are repulsed by them. To demonstrate actuation, ionomer membranes were immersed in a solution of water and salt, between two electrodes. Applying a voltage across the electrodes caused the membrane to bend. The paper, "Solvation-Driven Electrochemical Actuation," is published in the American Physical Society's Physical Review Letters.
According to the model developed by Porfiri and his group, the voltage caused a current of positive ions toward the negative electrode. These ions entered the membrane from one side, along with the water molecules in their solvation shells. On the other side of the membrane, positive ions and their solvation shells were dragged outside. The membrane responded like a sponge: the side full of water expanded, while the side with less water shrank. This differential swelling produced the macroscopic bending of the membrane. Studying actuation with different ions helps understand this phenomenon, as different ions attract a different number of water molecules around them.
The discovery of macroscopic mechanical consequences of solvation paves the way for more research on membranes. The group expects applications in the field of electrochemical cells (batteries, fuel cells, and electrolyzers), which often rely on the membranes utilized in this study. These membranes also share similarities with natural membranes, such as cell membranes, on which the mechanical effects of solvation are largely unknown.
More information: Alain Boldini et al. Solvation-Driven Electrochemical Actuation, Physical Review Letters (2021). DOI: 10.1103/PhysRevLett.126.046001
Provided by NYU Tandon School of Engineering



















