Cellulose nanofibril-based encapsulation structure to control drug release
Intelligent hydrogels with multiple functions such as sustained drug release abilities with low burst release, antibacterial properties, and biocompatibility are highly desirable in the biomaterials fields, particularly for cellulose-based drug carriers.
"Despite the nanocellulose-based hydrogels, which are promising for drug release applications, the issue of burst release at the beginning of releasing still needs to be addressed, and the drug release time still needs to be further improved," said by Associate Professor Li Bin, from the Qingdao Institute of Bioenergy and Bioprocess Technology (QIBEBT) of the Chinese Academy of Sciences.
Recently, the Metabolomics Group led by Prof. Cui Qiu from QIBEBT, in collaboration with researchers from Tianjin University of Science & Technology, constructed a new drug loading system with an encapsulation structure for controllable drug release. The study was published in ACS Applied Materials & Interfaces on Dec. 8.
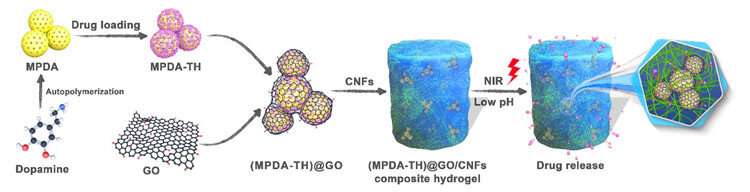
A well-known broad-spectrum antibiotic with low toxicity, tetracycline hydrochloride (TH), was used as the model drug. The researchers conducted physical crosslinking of mesoporous polydopamine (MPDA) nanoparticles wrapped with graphene oxide (GO) in cellulose nanofibril (CNF) hydrogels. They obtained a novel MPDA@GO/CNFs composite hydrogel with multiple responses, good mechanical strength and biocompatibility for controlled drug release.
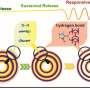
In this encapsulation structure, GO was used to package MPDA nanoparticles for prolonging drug release time, reducing burst release of the drug, and reinforcing physical strength of the obtained hydrogels. The sustained and controlled drug release behaviors of the composite hydrogels were highly dependent on pH value, and the rate of drug release could be accelerated by near infrared light irradiation.
"Importantly, this encapsulation structure shows good biocompatibility, and the toxicity of GO can be well shielded by CNFs hydrogels," said Prof. Li. "Longer drug release time and lower burst release can be achieved compared to the reported carriers for the same drug of TH."
This novel design of the CNF-based encapsulation structure will be beneficial to the development of new intelligent drug loading materials, and has potential applications for chemical and physical therapies.
More information: Yingying Liu et al. Construction of a Mesoporous Polydopamine@GO/Cellulose Nanofibril Composite Hydrogel with an Encapsulation Structure for Controllable Drug Release and Toxicity Shielding, ACS Applied Materials & Interfaces (2020). DOI: 10.1021/acsami.0c15465
Journal information: ACS Applied Materials and Interfaces
Provided by Chinese Academy of Sciences