November 5, 2020 report
Liquid-liquid phase separation found to drive the process of converting spidroin proteins to spider web fibers
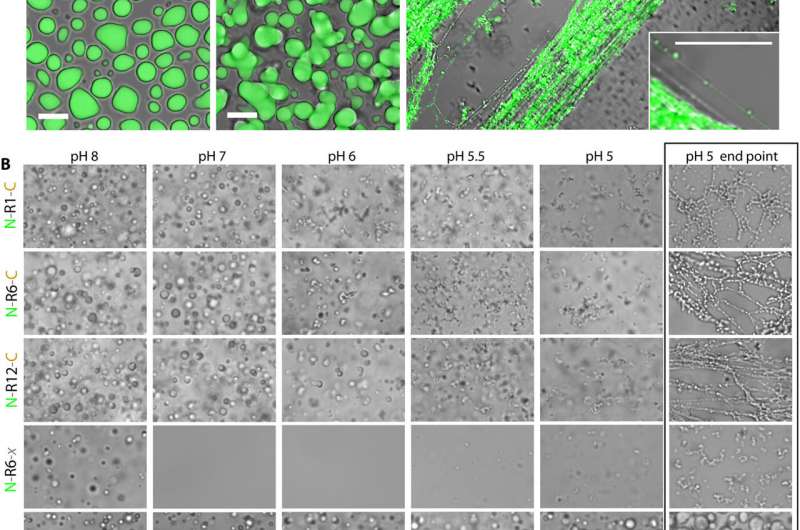
A team of researchers from the RIKEN Center for Sustainable Resource Science, Keio University and Kyoto University, has found that liquid-liquid phase separation (LLPS) drives the process of converting spidroin proteins to spider web fibers. In their paper published in the journal Science Advances, the group describes replicating the process of converting an internal protein to spider web fibers in their lab and how doing so allowed them to create strands of silk.
Scientists have been studying spiders and the webs they create for many years, with the goal of replicating the process and thereby industrializing the production of silk. Unfortunately, despite much effort, scientists still do not know how spiders do it. They do know that spiders create spidroin proteins, which they keep in a sac until they are ready to make a web. Then, as they eject the liquid proteins, the material somehow automatically forms into fibers to create strands. In this new effort, the researchers have found a way to replicate one part of this process.
The work involved engineering bacteria to produce the kind of spidroin proteins normally produced by the Joro spider. In studying the protein, they discovered that it grew cloudy when exposed to warm temperatures. A closer look showed the protein was coalescing and fusing, a likely first step toward the process of fiber formation. They also found that the process involved LLPS.
The team next tried exposing the protein to different materials until they hit on phosphate—adding it also made the protein cloudy. The team then began tuning the protein mix by altering pH levels until the mix began to create fibers. They next found that to make a strand, all they had to do was stretch the fiber-laden material. Doing so allowed them to create a strand 10 centimeters long. Studying the strand under a microscope showed it to have the same structure as strands produced naturally by the spiders.
The researchers note their work is just a tentative step toward fully understanding the process by which spiders create silk. They plan to push forward with their research with the ultimate goal of developing a way to produce silk in a lab, or a factory.
More information: Ali D. Malay et al. Spider silk self-assembly via modular liquid-liquid phase separation and nanofibrillation, Science Advances (2020). DOI: 10.1126/sciadv.abb6030
Journal information: Science Advances
© 2020 Science X Network





















