Creating perfect edges in 2-D-materials

Ultrathin materials such as graphene promise a revolution in nanoscience and technology. Researchers at Chalmers University of Technology, Sweden, have published a study in Nature Communications in which they present a method for controlling the edges of two-dimensional materials using a "magic" chemical.
"Our method makes it possible to control the edges—atom by atom—in a way that is both easy and scalable, using only mild heating together with abundant, environmentally friendly chemicals, such as hydrogen peroxide," says Battulga Munkhbat, a postdoctoral researcher at the Department of Physics at Chalmers University of Technology, and first author of the paper.
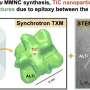
Materials as thin as just a single atomic layer are known as two-dimensional, or 2-D, materials. The most well-known example is graphene, as well as molybdenum disulphide, its semiconductor analog. Future developments within the field could benefit from studying one particular characteristic inherent to such materials—their edges. Controlling the edges is a challenging scientific problem, because they are very different in comparison to the main body of a 2-D material. For example, a specific type of edge found in transition metal dichalcogenides (known as TMD's, such as the aforementioned molybdenum disulphide), can have magnetic and catalytic properties.
Typical TMD materials have edges which can exist in two distinct variants, known as zigzag or armchair. These alternatives are so different that their physical and chemical properties are not at all alike. For instance, calculations predict that zigzag edges are metallic and ferromagnetic, whereas armchair edges are semiconducting and non-magnetic. Similar to these remarkable variations in physical properties, one could expect that chemical properties of zigzag and armchair edges are also very different. If so, it could be possible that certain chemicals might dissolve armchair edges, while leaving zigzag ones unaffected.
Now, such a magic chemical is exactly what the Chalmers researchers have found—in the form of ordinary hydrogen peroxide. At first, the researchers were completely surprised by the new results.
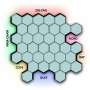
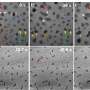
"It was not only that one type of edge was dominant over the others, but also that the resulting edges were extremely sharp—nearly atomically sharp. This indicates that the 'magic' chemical operates in a so-called self-limiting manner, removing unwanted material atom-by-atom, eventually resulting in edges at the atomically sharp limit. The resulting patterns followed the crystallographic orientation of the original TMD material, producing beautiful, atomically sharp hexagonal nanostructures," says Battulga Munkhbat.
"An extremely fascinating development"
The new method, which includes a combination of standard top-down lithographic methods with a new anisotropic wet etching process, therefore makes it possible to create perfect edges in two-dimensional materials.
"This method opens up new and unprecedented possibilities for van der Waals materials (layered 2-D materials). We can now combine edge physics with 2-D physics in one single material. It is an extremely fascinating development," says Timur Shegai, Associate Professor at the Department of Physics at Chalmers and leader of the research project.
These and other related materials often attract significant research attention, as they enable crucial advances within in nanoscience and technology, with potential applications ranging from quantum electronics to new types of nano-devices. These hopes are manifested in the Graphene Flagship, Europe's biggest ever research initiative, which is coordinated by Chalmers University of Technology.
To make the new technology available to research laboratories and high-tech companies, the researchers have founded a start-up company that offers high quality atomically sharp TMD materials. The researchers also plan to further develop applications for these atomically sharp metamaterials.
More information: Battulga Munkhbat et al, Transition metal dichalcogenide metamaterials with atomic precision, Nature Communications (2020). DOI: 10.1038/s41467-020-18428-2
Journal information: Nature Communications
Provided by Chalmers University of Technology