Breakthrough in graphene research: Large, stable pieces of graphene produced with unique edge pattern
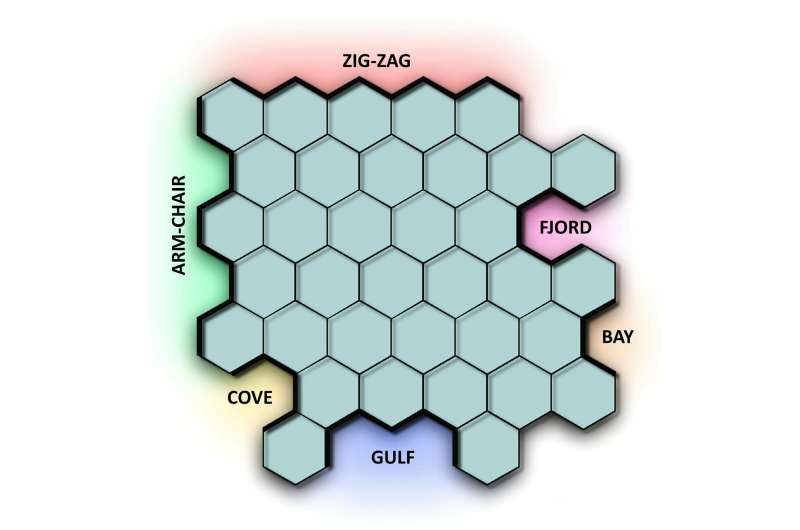
Bay, fjord, cove, armchair and zigzag—chemists use terms such as these to describe the shapes taken by the edges of nanographene. Graphene consists of a single-layered carbon structure in which each carbon atom is surrounded by three others. This creates a pattern reminiscent of a honeycomb, with atoms in each of the corners. Nanographene is a promising candidate to bring microelectronics down to the nano-scale and a likely substitute for silicon.
The electronic properties of the material depend greatly on its shape, size and above all, periphery—in other words how the edges are structured. A zigzag periphery is particularly suitable—in this configuration, the electrons, which act as charge carriers, are more mobile than in other edge structures. This means that using pieces of zigzag-shaped graphene in nanoelectronics components may allow higher frequencies for switches.
Materials scientists who want to research only zigzag nanographene confront the problem that this form makes the compounds unstable and difficult to produce in a controlled manner. This is a prerequisite, however, if the electronic properties are to be investigated in detail.
Researchers led by Dr. Konstantin Amsharov from the Chair of Organic Chemistry II have now succeeded in doing just that. Their research has now been published in Nature Communications. Not only have they discovered a straightforward method for synthesising zigzag nanographene, their procedure delivers a yield of close to 100 percent and is suitable for large scale production. They have already produced a technically relevant quantity in the laboratory.

The researchers first produced preliminary molecules, which they then fit together in a honeycomb formation over several cycles in a process known as cyclisation. In the end, graphene fragments are produced from staggered rows of honeycombs or four-limbed stars surrounding a central point of four graphene honeycombs, with the sought-after zigzag pattern at the edges. The product crystallises directly, even during synthesis. In their solid state, the molecules are not in contact with oxygen. In solution, however, oxidation causes the structures to disintegrate quickly.
This approach allows scientists to produce large pieces of graphene, while maintaining control over their shape and periphery. This breakthrough in graphene research means that scientists should soon be able to produce and research a variety of interesting nanographene structures, a crucial step toward using the material in nanoelectronic components.
More information: Dominik Lungerich et al, Dehydrative π-extension to nanographenes with zig-zag edges, Nature Communications (2018). DOI: 10.1038/s41467-018-07095-z
Journal information: Nature Communications
Provided by University of Erlangen-Nuremberg



















