Scientists propose nano-confinement strategy to form sub-nanometer reactors
Single-atom catalysts (SACs) are promising in electrocatalysis processes due to their maximum utilization of active species.
However, manipulation of these atomic-scale active sites to satisfy specific reactions is still an essential bottleneck due to their isolation features.
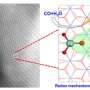
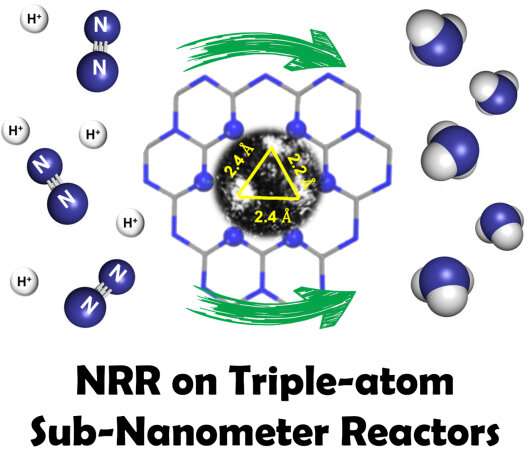
Prof. Liu Jian from the Dalian Institute of Chemical Physics (DICP) of the Chinese Academy of Sciences and his collaborators proposed a nano-confinement strategy to host multiple Fe and Cu single atoms inside the extremely narrow yet regular surface cavities of graphitic carbon nitride to form "sub-nanometer reactors."
The study was published in Advanced Materials on Sept. 2.
"These Fe and Cu atoms, highly confined in the sub-nanometer reactors, not only provide stronger interaction with the reactants but also, more importantly, lead to significant synergetic effect due to their unique microenvironments in this extremely narrow space, which is highly favorable for catalysis, especially the tandem processes such as the nitrogen reduction reaction," said Prof. Liang Ji from Tianjin University, a co-author of the study.
"This is the first time that we successfully and conceptually push the nanoreactors towards a much smaller dimension to form sub-nanometer reactors, which brings distinctively different properties from the conventional nanoreactors," added by Prof. Liu.
"First principle simulation reveals that this synergistic effect originates from the unique Fe-Cu coordination, which effectively modifies N2 absorption, improves electron transfer, and offers extra redox couples for nitrogen reduction reaction," said Prof. Sun Chenghua from Swinburne University of Technology, another co-author of the study.
The researchers found that this significant synergy caused by the multiple confined atoms led to significant performance enhancement for the model electrocatalytic process, the nitrogen reduction reaction (NRR).
Improvements in terms of high ammonia yield and efficiency that are much higher in comparison with the mono-metal counterparts have been achieved.
This concept of constructing sub-nanometer reactors not only provides a new strategy of manipulating catalysts active centers at the subnanometer scale, but also sheds light on the design of novel catalysts with a precision spatial location at the sub-nanometer scale for a wide spectrum of catalytic reactions as well.
More information: Xiaowei Wang et al. Confined Fe–Cu Clusters as Sub‐Nanometer Reactors for Efficiently Regulating the Electrochemical Nitrogen Reduction Reaction, Advanced Materials (2020). DOI: 10.1002/adma.202004382
Journal information: Advanced Materials
Provided by Chinese Academy of Sciences