Researchers develop non-precious alloy catalyst for cinnamaldehyde
Recently, scientists at the Institute of Solid State Physics, Hefei Institutes of Physical Science developed a non-precious alloy catalyst with excellent catalytic activity, selectivity and stability for selective hydrogenation (SH) of cinnamaldehyde (CAL).
Cinnamyl alcohol (COL) has been an essential compound widely used in the flavoring, perfume and pharmaceutical industries. But SH of CAL to COL is challenged by the unfavored thermodynamics that leads to low selectivity toward COL. And the use of an organic solvent in COL production pollutes the environment and requires post-production separation.
To improve the selectivity of CAL hydrogenation, transition-metal/noble-metal (Pt is the most used) bimetallic catalysts is employed to create synergetic electronic effects that thermodynamically favored the hydrogenation of C=O in CAL. However, the use of scarce and expensive noble-metals is undesirable.
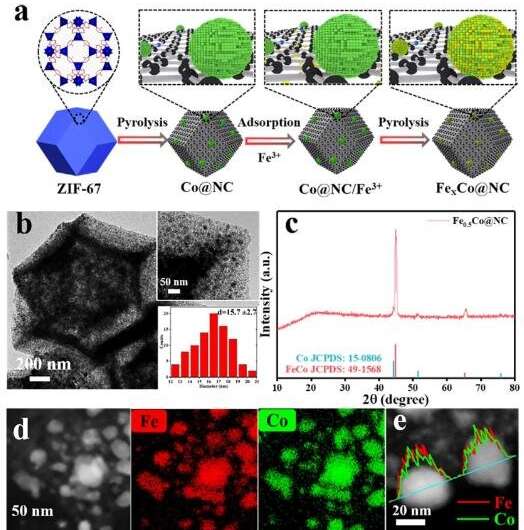
In this study, the researchers reported that, for the first time, a solution-based metal ion impregnation approach was innovatively combined with pyrolysis to achieve the controllable synthesis of highly dispersed Fe-Co alloy nanoparticles (NPs) on N-doped graphitic carbon support.
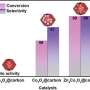
They stated that the COL selectivity and CAL conversion efficiency were respectively promoted by the presence of Co and Fe, while the synergism of the alloyed Fe-Co was the key to concurrently achieve high COL selectivity and CAL conversion efficiency.
Another finding was that the H2O played an important role in CAL conversion. For CAL conversation, the higher the solvent polarity, the higher the CAL conversion efficiency. For COL selectivity, it was categorically confirmed that the promotional effect of H2O solvent remarkably enhanced the hydrogenation on C=O and concurrently suppressed the hydrogenation on C=C.
More information: Fe‐Co Alloyed Nanoparticles Catalyzed Efficient Hydrogenation of Cinnamaldehyde to Cinnamyl Alcohol in Water. Angew. Chem. Int. Ed. doi.org/10.1002/anie.202009913
Journal information: Angewandte Chemie International Edition
Provided by Chinese Academy of Sciences