New study finds two amino acids are the Marie Kondo of molecular liquid phase separation

The last several years have brought mounting evidence that the molecules inside our cells can self-organize into liquid droplets that merge and separate like oil in water in order to facilitate various cellular activities. Now, a team of biologists at the Advanced Science Research Center at The Graduate Center, CUNY (CUNY ASRC) have identified unique roles for the amino acids arginine and lysine in contributing to liquid phase properties and their regulation. Their findings are available today online in Nature Communications.
Known as liquid-liquid phase separation, the process allows some molecules within a cell to cloister themselves into membraneless organelles in order to carry out certain duties without interruption from other molecules. The mechanism can also allow molecules to create multiphase droplets that resemble, say, a drop of honey inside a drop of oil surrounded by water in order to carry out sophisticated jobs.
"This is a really exciting new research area because it uncovers a core biological function that, when gone awry, may be at the root of disease, particularly neurodegeneration as in ALS or Alzheimer's," said principal investigator and Graduate Center, CUNY Biochemistry Professor Shana Elbaum-Garfinkle, whose lab at the CUNY ASRC Structural Biology Initiative conducted the study. "With an understanding of how individual amino acids contribute to phase behavior, we can begin to investigate what's going wrong in liquid phase separation that may interfere with normal biological function and potentially design therapies that can modulate the process."
Researchers have suspected for a while that arginine and lysine—two of the 20 amino acids that make up all proteins—were responsible for regulating liquid phase separation, but they weren't certain how each contributed to phase behavior and to creating the differing viscosities that cloister molecules into separate communities.
"Arginine and lysine are very similar amino acids in terms of both being positively charged, but they differ in terms of binding capability. We were really curious to understand what effect this difference would have on the material properties, such as viscosity or fluidity, of the droplets they form," said Rachel Fisher, the paper's first author and a postdoc in Elbaum-Garfinkle's lab. "We also wanted to know how these differences manifest themselves when the arginine and lysine systems are combined. Will the droplets coexist? When we saw they did, we then wanted to understand how we could modulate this multi-phase behavior."
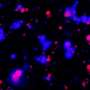
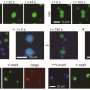
To answer their questions, Elbaum-Garfinkle's team used a technique called microrheology—whereby tiny tracers are used to probe material structures—to track and investigate the properties of arginine and lysine droplets. They found that arginine-rich droplets were over 100 times more viscous than lysine-rich droplets, comparable to the difference between a thick syrup or ketchup and oil. The viscosity differences are significant enough that if lysine and arginine polymers are combined, they don't mix. Instead, they create multi-phase droplets that sit within one another like Dutch nesting dolls. Additionally, arginine has such strong binding properties that under some conditions it can compete with lysine and replace or dissolve lysine droplets. The researchers further identified ways to tune the balance between competition and coexistence of the two phases. The results present a novel mechanism for designing, controlling or intervening in molecular liquid phases.
More information: Nature Communications, DOI: 10.1038/s41467-020-18224-y
Journal information: Nature Communications
Provided by CUNY Advanced Science Research Center