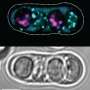
Survival of the fit-ish

It can be hard to dispute the common adage 'survival of the fittest.' After all, "most of the genes in the genome are there because they're doing something good," says Sarah Zanders, Ph.D., assistant investigator at the Stowers Institute for Medical Research. But, she says, "others are just there because they've figured out a way to be there."
The conventional understanding of evolution is that genes encoding a beneficial function are the most frequently transmitted, which ensures that the fittest organisms—the ones that have traits most favorable for their environment—survive. Less known is the fact that there exist parasitic gene elements within an organism that are doing just the opposite.
"The way one could think of it is that the genome is like a society," explains Zanders. "Within that society, there are individuals who derive their living from doing good things and making valuable contributions. But there are others who don't contribute in beneficial ways and are actually harmful to society," explains Zanders.
The Zanders Lab studies parasitic genes in Schizosaccharomyces pombe, a species of fission yeast sometimes found in the popular fermented tea drink kombucha. Because of its simplicity and fast generation times, S. pombe is a highly tractable system for studying parasitic gene elements, particularly a class known as meiotic drivers. Instead of conferring a benefit to their host, meiotic drivers confer distinct disadvantages to their host, in order to bias their own perpetuation. One method of doing so, employed by the wtf meiotic drive genes, is through selective poisoning of cells in meiosis.
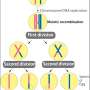
Meiosis is the process of cell division through which sexually-reproducing organisms form gametes—such as egg and sperm cells in humans, or spores in yeast—to propagate the next generation. Normally, this process results in gametes that inherit one of two copies of each chromosome carried by the parent cell, and each copy is transmitted to gametes at an equal rate. Meiotic drivers, however, short-circuit this law of Mendelian segregation.
"Usually all the alleles—or variants of a particular gene—get a fair chance, and natural selection can pick the best ones," explains Zanders. "But alleles that are meiotic drivers select themselves even if they're not the best option. And they're never the best option."
In a paper published online August 13, 2020, in eLife, members of the Zanders lab explain how it could be possible that meiotic drivers persist in the population, even as they kill off many would-be hosts. It turns out that S. pombe can employ variants of other genes to help suppress the negative effect of meiotic drivers, albeit at a cost to fitness.
Usually, to propagate laboratory strains of S. pombe, they are inbred. "When you outcross two isolates that are almost identical, they produce almost no progeny," explains first author María Angélica Bravo Núñez, Ph.D., who was involved in the identification of the wtf genes, and who did this work in the Zanders Lab as a predoctoral researcher at the Graduate School of the Stowers Institute. "This suggested there might be some type of competition at play." Bravo Núñez and colleagues used this reasoning to seek out genes that could be in conflict in similar, but non-identical, genomes of S. pombe. The central experimental design of the current work involved outcrossing of S. pombe isolates that are over 99% identical.
"Outcrossing can have many advantages," says Bravo Núñez, such as providing a normal allele of a gene to rescue the effect of a mutant allele. "But the meiotic drive genes that we study actually exert their deleterious effect in the heterozygous scenario, where the alleles of a gene are not the same."
One illuminating experiment they did was to compare outcomes of inbred and outcrossed S. pombe isolates in a genetic background containing a mutation in rec12, which is a gene that promotes proper chromosome segregation.
"Rec12 usually promotes fertility," says Bravo Núñez. "When we removed rec12 function, the numbers of viable spores decreased, but only in the inbred scenario. The relative numbers of viable spores didn't really decrease in the outcrossed scenario." Moreover, of the viable spores, they found that survival was biased towards atypical gamete products, resulting from mis-segregation and unequal crossover of paired chromosomes. In other words, surviving spores contained the maximal number of meiotic drivers, sometimes due to inheriting an extra chromosome. This finding was surprising because extra chromosomes are usually considered very harmful. In humans, for instance, extra or missing chromosomes can lead to inviable gametes, severe birth defects, or infertility.
"When you have heterozygosity of wtf genes, making a mutation in the rec12 gene is relatively good for the organism, because that's just what has to happen in order to not kill all the progeny," says Bravo Núñez.
"We think of rec12 as this gene that's really important for fertility," explains Zanders. "But the situation is totally different when there is outcrossing. Having rec12 doesn't actually help at all, because the yeast is better off making gametes that do not result from proper chromosome segregation. This is just one example of the power of meiotic drivers to change the landscape of meiosis. Fundamentally, what is 'good' for the organism has changed."
"Having that extra chromosome is not good, and the yeast colonies look unusual, small, and irregular. But after they continue to grow for a while, the cells lose that extra chromosome and then they can thrive as haploids. So, this step is actually just temporary," says Bravo Núñez.
Precisely because meiotic drivers exert their influence in a heterozygous scenario, they are easy to miss. "There are many flavors of meiotic drive. Some forms of meiotic drive are hard to measure experimentally because the bias is so subtle," says Zanders. "We're not the first to study meiotic drivers in depth. We just have a better model system now, so we can make more progress faster."
"Drive systems tend to be repetitive, and you can usually find them in various copies in genomes," says Bravo Núñez. "They have, in many cases, already been found in other systems, such as fungi, mice, and fruit flies, but are not yet fully characterized." The study of meiotic drivers in S. pombe "will hopefully guide future research to understand other drive systems."
"Humans certainly have meiotic drive genes. Whether or not they have meiotic drive genes of the gamete-killing type is unclear," says Zanders. "Meiotic drive has likely affected the evolution of human centromeres, which are regions of chromosomes that are very important for proper chromosome segregation. Certain chromosome fusions exhibit meiotic drive in humans, as do sequences that are involved in DNA recombination. We're going to continue focusing on these and other parasitic gene elements, their strategies, and their effects."
Other coauthors of this work include Ibrahim M. Sabbarini and Lauren E. Eide from the Stowers Institute and Robert L. Unckless, Ph.D., from the University of Kansas.
This work was funded in part by the Stowers Institute for Medical Research, March of Dimes Foundation Basil O'Connor Starter Scholar Research Award (5-FY18-58 to SEZ), Searle Scholar Award (to SEZ), and National Institutes of Health (National Institute of General Medical Sciences awards R00GM114436 and DP2GM132936 to SEZ and National Cancer Institute award F99CA234523 to MABN). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Lay Summary of Findings
A new study published online August 13, 2020, in eLife from the laboratory of Sarah Zanders, Ph.D., at the Stowers Institute for Medical Research describes a strategy that the S. pombe fission yeast genome can use to mitigate some of the worst effects conferred by parasitic gene elements known as meiotic drivers.
The meiotic drivers analyzed in the study are able to short-circuit the conventional law of Mendelian segregation, which usually ensures that each gamete (reproductive cell) receives one of two copies of each chromosome from the parent cell with equal transmission. Meiotic drivers instead can poison gametes that do not contain their genetic sequence, thereby swinging the transmission rate in their favor.
As a way of enabling the survival of some gametes that would otherwise die, the S. pombegenome can employ variants of other genes to create a situation that "protects" them, even if it comes at a cost to fitness. For example, a mutation in rec12, a gene responsible for proper chromosome segregation, can lead to gametes having extra chromosomes. While extra chromosomes are usually undesirable, in this scenario they actually enable more gametes to survive. This surprising insight into meiotic drivers and how they can be suppressed could help researchers better understand the forces that shape the evolution of gamete formation, as well as those underlying human infertility.
More information: María Angélica Bravo Núñez et al. Atypical meiosis can be adaptive in outcrossed Schizosaccharomyces pombe due to wtf meiotic drivers, eLife (2020). DOI: 10.7554/eLife.57936
Journal information: eLife
Provided by Stowers Institute for Medical Research