July 27, 2020 feature
Designer nanozymes for reactive-oxygen species scavenging anti-inflammatory therapy
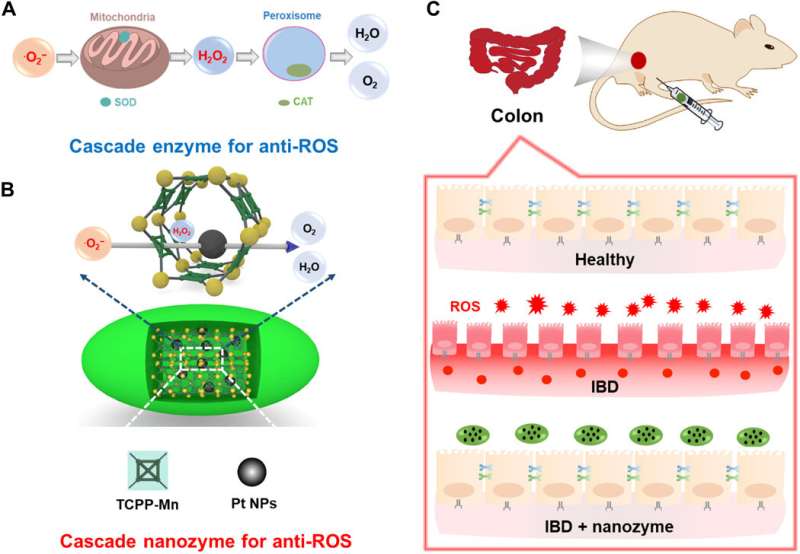
In a recent report, Yufeng Liu and a team of interdisciplinary researchers in China developed an integrated nanozyme cascade to eliminate excessive reactive oxygen species (ROS; oxygen free radicals). The nanozyme mimicked superoxide dismutase (a group of enzymes) and incorporated a manganese (Mn)-based metal-organic framework (MOF) to transform oxygen radicals to hydrogen peroxide (H2O2). Using in-lab and in vivo experiments, the team showed the ROS-scavenging potential of integrated cascade nanozymes. As proof of concept, they relieved two forms of inflammatory bowel disorder (IBD)—ulcerative colitis and Crohn's disease using cascade nanozymes as effective treatments. The study provided a new method to construct enzyme-like cascade systems and illustrate the promise of their efficient therapy to treat IBD in vivo. The work is now published on Science Advances.
During cascade catalytic reactions that occur in living organisms, multiple enzymes are combined inside subcellular compartments for accurate signal transduction and effective metabolism. Such confined cascade reactions are advantageous compared to conventional multi-step reactions due to reduced diffusion barriers and enhanced local concentrations of intermediates for improved atom economy and reactions in total. Scientists have made substantial efforts to mimic such cascade systems on scaffolds, but their high cost and low stability have limited broad practical applications. As a result, researchers have explored enzyme mimics to understand and construct cascade reactions in the lab. In this work, Liu e al. detailed a single-component nanozyme-based cascade reaction system with high activity and demonstrated their role during the in vivo therapy of reactive oxygen species (ROS)-associated inflammatory bowel disease (IBD).
Design, synthesis and characterization of the enzyme scaffold
The team designed and synthesized an integrated superoxide dismutase (SOD) and catalase (CAT) mimetic cascade nanozyme and designated it as Pt@PCN222-Mn. To construct the scaffold, they introduced a SOD-like moiety of Manganese (Mn) (III) porphyrin and a CAT-like platinum nanoparticle within a nanoscale Zirconium (Zr)-based metal organic framework (MOF) named PCN222. The integrated construct (Pt@PCN222-Mn) showed enhanced ROS-scavenging activity to protect mice from ROS-related IBD, an incurable chronic disease as yet, and thereby to broaden the potential of cascade nanozymes for in vivo biomedical applications.

During the experiments, Liu et al. confirmed the successful synthesis of the preliminary manganese based constructs using nuclear magnetic resonance, Fourier transform infrared spectroscopy and ultraviolet visible spectroscopic measurements. The team synthesized the component containing a nanoscale MOF and confirmed its formation with transmission electron microscopy (TEM) and scanning electron microscopy (SEM). The scientists then developed three cascade nanozymes and termed them Pt@PCN222-Mn-1, Pt@PCN222-Mn-3 and Pt@PCN222-Mn-5, respectively. Of these, Pt@PCN222-Mn-5 had a higher surface area compared to other anti-ROS nanozymes investigated thus far.
Investigating the catalytic activity of the cascade nanozymes
Scavenging for oxygen radicals (•O2−) is the initial step of the anti-ROS cascade reaction, therefore the team investigated the catalytic activity of the synthetic constructs to monitor their capability to similarly eliminate oxygen radicals. The work highlighted the key role of manganese in porphyrin to mimic superoxide dismutase (SOD)-like activities of the different enzyme constructs, where Pt@PCN222-Mn-5 constructs showed the highest SOD-like activity among the enzyme mimics. The team confirmed the results using electron paramagnetic resonance (EPR) spectroscopy.
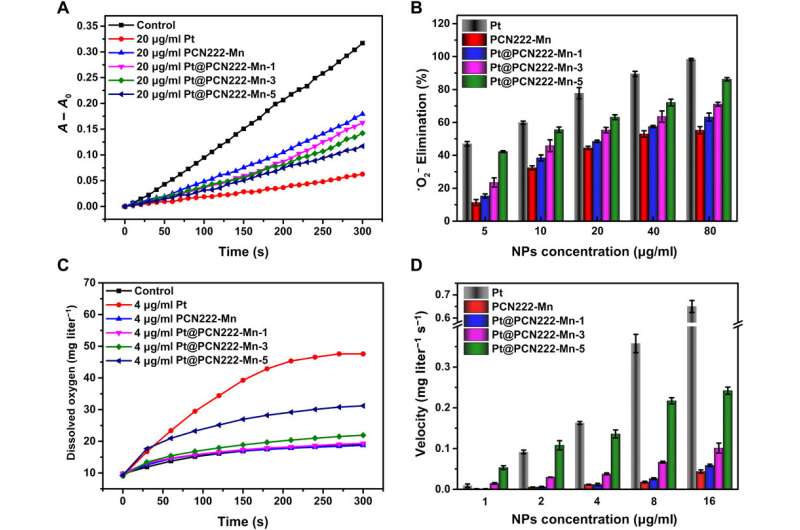
As a second crucial step in the ROS-scavenging cascade in biological environments, the CAT (catalase) enzyme catalyzed the decomposition of hydrogen peroxide (H2O2) into water and oxygen. Liu et al. therefore monitored the CAT-like activities of the nanozymes by monitoring oxygen generated from decomposing hydrogen peroxide. As before, Pt@PCN222-Mn-5 showed the highest CAT-like activity among MOF-based enzyme mimics. Based on the results, Liu et al. confirmed the SOD-like activity to result from the manganese-bound moiety of the enzyme construct, while integral platinum nanoparticles were mainly responsible for the CAT-like activity.
Focus enzyme Pt@PCN222-Mn-5 and in vivo anti-inflammatory therapy
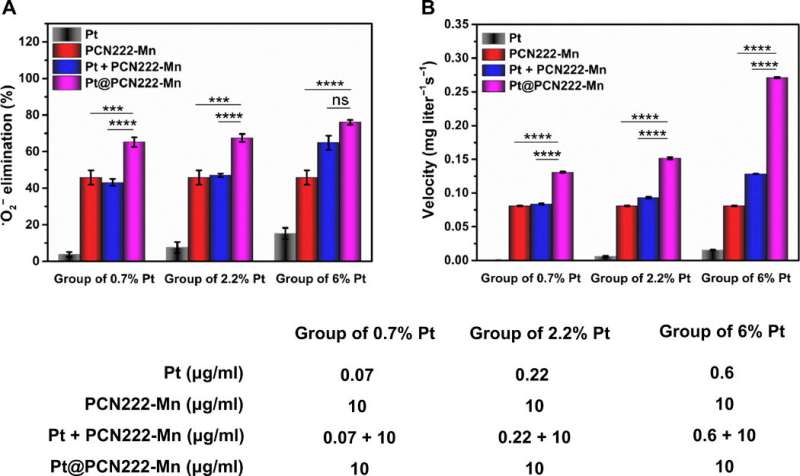
To fully comprehend the activity of the cascade nanozyme Pt@PCN222-Mn-5, the team investigated its synergistic SOD-like and CAT-like enzymatic activity and observed pronounced synergistic effects for the latter. The team chose the nanozyme for further investigations and tested its functionality with a cell line during cytotoxicity studies. At a concentration below 80 µg/mL, the nanozyme did not show any cytotoxicity and demonstrated excellent reactive oxygen species scanning potential.
Based on the experimental results, Liu et al. conducted in vivo therapies to treat ulcerative colitis (UC) and Crohn's disease (CD) in mouse models of the disease. Since UC is a form of inflammatory bowel disorder that is typically accompanied with overproduced reactive oxygen species, the scientists tested the therapeutic effects of Pt@PCN222-Mn-5 together with existing medication for IBD treatment. The team administered the treatment via intraperitoneal injection to each mouse and observed successful therapeutic effects of Pt@PCN222-Mn-5 to relieve the UC disease state in mice. The team optimized the doses of the cascade nanozyme using several treatment groups and found the focus enzyme of interest to be the most favorable. When they similarly explored CD, another type of IBD using a disease-induced mouse model, the treatment strategy indicated greater efficacy for cascade nanozymes compared to traditional small-molecule medications used in IBD treatment.
![LEFT: UC therapy with Pt@PCN222-Mn-5. (A) Overall procedure of the animal experiment (DSS-induced colitis). (B) Daily body-weight development for 10 days. (C) Changes in body weight of mice before (day 7) and after indicated treatments (day 10). (D) Images of the colons and (E) the corresponding colon lengths in indicated groups. (F) IL-1β and (G) TNF-α levels in colon homogenates from indicated groups. (H) H&E-stained colonic sections of mice from indicated groups on day 10. The data are shown as means ± SD (n = 5). *P < 0.05, **P < 0.01, ***P < 0.005, and ****P < 0.001; ns, not significant; t test. Photo credit (D): Yufeng Liu, College of Engineering and Applied Sciences, Nanjing University. RIGHT: CD therapy with Pt@PCN222-Mn-5. (A) Overall procedure of the animal experiment [2,4,6-trinitro benzene sulfonic acid (TNBS)–induced colitis]. (B) Daily body-weight development after administering TNBS solution into the colon lumen. (C) Changes in body weight of mice before (day 9) and after indicated treatments (day 12). (D) Colon lengths and (E) the corresponding images of colons of indicated groups. The data are shown as means ± SD (n = 5). *P < 0.05, ***P < 0.005, and ****P < 0.001; ns, not significant; t test. Photo credit (E): Yuan Cheng, College of Engineering and Applied Sciences, Nanjing University. Credit: Science Advances, doi: 10.1126/sciadv.abb2695 Designer nanozymes for reactive-oxygen species scavenging anti-inflammatory therapy](https://scx1.b-cdn.net/csz/news/800a/2020/4-designernano.jpg)
In this way, Yufeng Liu and colleagues developed an integrated nanozyme to catalyze cascade reactions and eliminate reactive oxygen species (ROS). The integrated nanozyme had two spatially separated active sites to mimic superoxide dismutase (SOD) and catalase (CAT). Using in-lab experiments they showed the outstanding ROS-scavenging activity of the cascade nanozyme, their excellent biocompatibility and good aqueous dispersibility. The team established an inflammatory animal model to test the anti-ROS capacity of the cascade nanozyme in a biological organism and determined their superior therapeutic potential towards both ulcerative colitis (UC) and Crohn's disease (CD) models of inflammatory bowel disorder (IBD). The team optimized the concentration of the nanozyme to achieve effective relief of IBD in animal models. The work showed excellent ROS-scavenging activities for inflammatory treatment and provides a promising method to construct nanozymes with multiple active sites for further applications in biomedicine.
More information: Yufeng Liu et al. Integrated cascade nanozyme catalyzes in vivo ROS scavenging for anti-inflammatory therapy, Science Advances (2020). DOI: 10.1126/sciadv.abb2695
Wei-Hai Chen et al. Biocatalytic cascades driven by enzymes encapsulated in metal–organic framework nanoparticles, Nature Catalysis (2018). DOI: 10.1038/s41929-018-0117-2
Stefan Wirtz et al. Chemically induced mouse models of intestinal inflammation, Nature Protocols (2007). DOI: 10.1038/nprot.2007.41
Journal information: Science Advances , Nature Protocols , Nature Catalysis
© 2020 Science X Network