Scientists propose novel method to synthesize atomic dispersed Cu-N-C catalyst
Non-noble metal nitrogen doped carbon material (M-N-C) catalysts are considered as potential alternatives for precious metals, due to their abundance, biocompatibility, environmental benign and high catalytic performance.
However, the current synthetic strategies for fabricating M-N-C catalysts via high-temperature pyrolysis unavoidably lead to a structural heterogeneity.
In order to obtain M-N-C with atomically dispersed active sites, a post-treatment is required to remove the inorganic metal-containing nanoparticles. This makes the whole synthesis complicated and environmentally unfriendly, and even damages the structure of M-Nx site.
Recently, a research team led by Prof. YANG Yong from the Qingdao Institute of Bioenergy and Bioprocess Technology (QIBEBT) of the Chinese Academy of Sciences has developed a simple, economical and efficient high-temperature pyrolysis synthetic method. The study was published in ACS Applied Materials & Interfaces.
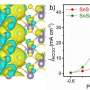
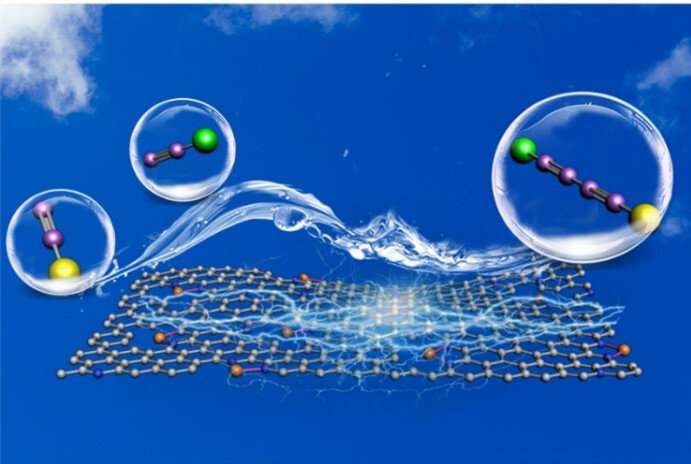
Using environmentally friendly biochar and inexpensive Cu (NO3)2 as the raw materials without post-treatment process, scientists have synthesized the atomic dispersed Cu-N-C catalyst. The atomic dispersion of coordinatively unsaturated Cu-N2 sites is on biomass-derived hierarchically N-doped porous carbon with high specific surface area.
"This catalyst exhibits outstanding catalytic performance for Glaser-Hay coupling of terminal alkynes under base-free, ligand-free conditions by using air as oxidant," said REN Peng, a postgraduate student from QIBEBT and first author of the study.
This protocol has addressed the poor selectivity issue in the synthesis of asymmetric 1,3-diyens, it's an important advancement to the field, commented by the peer reviewers.
Experimental results and theoretical calculations revealed that the low N coordination number of single-atom Cu sites in Cu-N2 exhibited a preferential adsorption to terminal alkyne; meanwhile, the adjacent pyridinic N sites on the carbon matrix facilitated the deprotonation of the adsorbed alkyne to generate the key intermediate species, thus synergistically boosting the reaction.
This work not only provides an alternative facile synthetic strategy for fabricating atomically dispersed M-N-C catalysts, but also represents a significant advance for accessing (un)symmetrical 1,3-diynes from Glaser-Hay coupling.
More information: Peng Ren et al. Facile Fabrication of the Cu-N-C Catalyst with Atomically Dispersed Unsaturated Cu-N2 Active Sites for Highly Efficient and Selective Glaser–Hay Coupling, ACS Applied Materials & Interfaces (2020). DOI: 10.1021/acsami.0c05100
Journal information: ACS Applied Materials and Interfaces
Provided by Chinese Academy of Sciences