New coating material for surgical tools
Spin-off company NChemi Engineering Nanomaterials, created from the Center for the Development of Functional Materials (CDMF) in Brazil, has presented new technology resulting from projects developed by the Universitat Jaume I (UJI) and the CDMF.
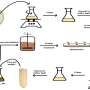
The product Plenus Duty is a coating for industrial applications that has the properties of increasing the hardness of a metal surface by 90% and reducing friction by 50%. This material is capable of forming a thin nanometric film of zirconium adhered to the surface of stainless steel that enhaces these two characteristics.
This spin-off company is an example of how, from multidisciplinary, basic and fundamental research, it is possible to simulate, model and predict the behaviour of new materials under different conditions at the micro and nano scale, thus achieving results at the forefront of knowledge with added know-how and giving rise to new technologies with a high socio-economic impact.
All this is the result of the international collaboration, maintained for over 30 years, between the Laboratory of Theoretical and Computational Chemistry of the UJI, coordinated by Juan Andrés Bort, and the CDMF, directed by Elson Longo.
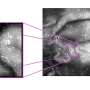
The technology based on the Plenus Med material is applied to instruments of any size and shape (without the need for any modification or pre-treatment) and maintains the sharpness of the pieces for longer amounts of itme (guarantees their stability and reliability during surgery); the final thickness of the coating is less than 0.2 microns and there are no losses in relation to sharpness, thus proving to be superior to diamond coating (DLC). In addition, it is made of non-toxic, biocompatible material.
The nanometre ceramic coating achieves a local reduction in tissue/bone necrosis and, consequently, a faster and less traumatic post-surgical recovery. It has been tested by different medical and surgical staff and has achieved a high approval rate. Furthermore, a surgical material manufacturer has obtained approval from Anvisa (the regulatory body in Brazil) to use it.
Provided by Asociacion RUVID