A synthetic genetic circuit to quantify repeat deletion in bacteria
Repeat sequences are ubiquitous in the genomes of prokaryotes and eukaryotes. The rearrangement between direct repeats can result in deletions or expansions of DNA sequences, contributing to the genetic plasticity, regulation of transcription and protein coding sequence variations.
Thus, quantification of a genetic rearrangement rate is a primary aspect in deciphering its underlying mechanisms.
The previous antibiotic resistance selection method, commonly achieved by inserting direct repeats within the coding sequence of tetR (resistance gene for tetracycline), has been used to quantify the direct repeat deletion (DRD) rate. But it was limited in false positive events and affected by physiological process from the antibiotic, and the length of repeat arms (LRA).
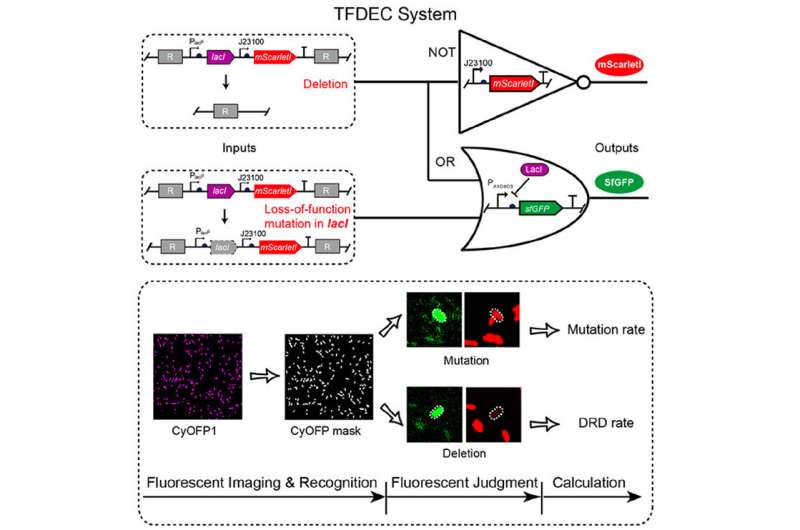
Scientists from the Shenzhen Institutes of Advanced Technology (SIAT) of the Chinese Academy of Sciences and Huazhong University of Science and Technology (HUST) have designed a synthetic genetic circuit, termed TFDEC, which stands for Three color Fluorescence-based Deletion Event Counter, to quantify the DRD rate under neutral conditions. The study was published in ACS Synthetic Biology.
They designed a fluorescent logic gate-based method to quantify the DRD rate by distinguishing the deletion and mutation events from all detected events with fluorescent intensity. They found that the DRD rate was RecA-dependent for long LRA (>500bp) and RecA-independent for short LRA (<500bp) in Pseudomonas aeruginosa.
They also found that the increase of DRD rate followed an S-shaped curve with the increase of length of repeat arm, while treating cells with ciprofloxacin enhanced the DRD rate but did not change the LRA-dependence of DRD. In addition, the DRD rate increased for long LRA in the uvrD (8-fold) and radA (4-fold) mutants, which was related to RecA protein.
This study may shed light on investigation of a persister emergence during antibiotic treatment and unravelling the roles of certain proteins during stress-driven evolutions of bacterial population.
More information: Yajia Huang et al. A Synthetic Genetic Circuit Enables Precise Quantification of Direct Repeat Deletion in Bacteria, ACS Synthetic Biology (2020). DOI: 10.1021/acssynbio.9b00256
Journal information: ACS Synthetic Biology
Provided by Chinese Academy of Sciences