Novel 3-D imaging technology makes fluorescence microscopy more efficient

Scientists have been using fluorescence microscopy to study the inner workings of biological cells and organisms for decades. However, many of these platforms are often too slow to follow the biological action in 3-D; and too damaging to the living biological specimens with strong light illumination.
To address these challenges, a research team led by Dr. Kevin Tsia, Associate Professor of the Department of Electrical and Electronic Engineering and Programme Director of Bachelor of Engineering in Biomedical Engineering of the University of Hong Kong (HKU), developed a new optical imaging technology—coded light-sheet array microscopy (CLAM) - which can perform 3-D imaging at high speed, and is power efficient and gentle enough to preserve living specimens during scanning at a level that is not achieved by existing technologies.
This advanced imaging technology was recently published in Light: Science & Applications. A US patent application has been filed for the innovation.
"CLAM allows 3-D fluorescence imaging at high frame rate comparable to state-of-the-art technology (~10's volumes per second). More importantly, it is much more power efficient, being over 1,000 times gentler than the standard 3-D microscopes widely used in scientific laboratories, which greatly reduces the damage done to living specimens during scanning," explained Dr. Tsia.
Existing 3-D biological microscopy platforms are slow because the entire volume of the specimen has to be sequentially scanned and imaged point-by-point, line-by-line or plane-by-plane. In these platforms, a single 3-D snapshot requires repeated illumination on the specimen. The specimens are often illuminated with thousands to million of times more intensity than that of sunlight. This is likely to damage the specimen itself, thus is not favorable for long-term biological imaging for diverse applications like anatomical science, developmental biology and neuroscience.
Moreover, these platforms often quickly exhaust the limited fluorescence "budget"—a fundamental constraint that fluorescent light can only be generated upon illumination for a limited period before it permanently fades out in a process called "photo-bleaching," which sets a limit to how many image acquisitions can be performed on a sample.

"Repeated illumination on the specimen not only accelerates photo-bleaching, but also generates excessive fluorescence light that does not eventually form the final image. Hence, the fluorescence 'budget' is largely wasted in these imaging platforms," Dr. Tsia added.
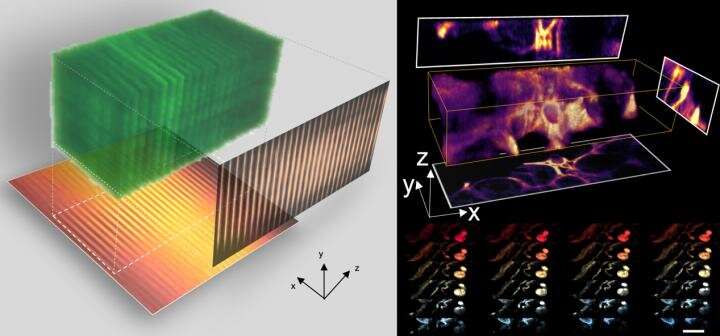
The heart of CLAM is transforming a single laser beam into a high-density array of 'light-sheets' with the use of a pair of parallel mirrors, to spread over a large area of the specimen as fluorescence excitation.
"The image within the entire 3-D volume is captured simultaneously (i.e. parallelized), without the need to scan the specimen point-by-point or line-by-line or plane-by-plane as required by other techniques. Such 3-D parallelization in CLAM leads to a very gentle and efficient 3-D fluorescence imaging without sacrificing sensitivity and speed," as pointed out by Dr. Yuxuan Ren, a postdoctoral researcher on the work. CLAM also outperforms the common 3-D fluorescence imaging methods in reducing the effect of photo-bleaching.
To preserve the image resolution and quality in CLAM, the team turned to Code Division Multiplexing (CDM), an image encoding technique which is widely used in telecommunication for sending multiple signals simultaneously.
"This encoding technique allows us to use a 2-D image sensor to capture and digitally reconstruct all image stacks in 3-D simultaneously. CDM has never been used in 3-D imaging before. We adopted the technology, which became a success," explained by Dr. Queenie Lai, another postdoctoral researcher who developed the system.
As a proof-of-concept demonstration, the team applied CLAM to capture 3-D videos of fast microparticle flow in a microfluidic chip at a volume rate of over 10 volumes per second comparable to state-of-the-art technology.
"CLAM has no fundamental limitation in imaging speed. The only constraint is from the speed of the detector employed in the system, i.e. the camera for taking snapshots. As high-speed camera technology continually advances, CLAM can always challenge its limit to attain an even higher speed in scanning," highlighted by Dr. Jianglai Wu, the postdoctoral research who initiated the work.
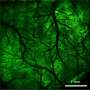
The team has taken a step further to combine CLAM with HKU LKS Faculty of Medicine's newly developed tissue clearing technology to perform 3-D visualization of mouse glomeruli and intestine blood vasculature in high frame-rate.
"We anticipate that this combined technique can be extended to large-scale 3-D histopathological investigation of archival biological samples, like mapping the cellular organization in brain for neuroscience research." Dr. Tsia said.
"Since CLAM imaging is significantly gentler than all other methods, it uniquely favours long term and continuous 'surveillance' of biological specimen in their living form. This could potentially impact our fundamental understanding in many aspects of cell biology, e.g. to continuously track how an animal embryo develops into its adult form; to monitor in real-time how the cells/organisms get infected by bacteria or viruses; to see how the cancer cells are killed by drugs, and other challenging tasks unachievable by existing technologies today," Dr. Tsia added.
CLAM can be adapted to many current microscope systems with minimal hardware or software modification. Taking advantage of this, the team is planning to further upgrade the current CLAM system for research in cell biology, animal and plant developmental biology.
More information: Yu-Xuan Ren et al, Parallelized volumetric fluorescence microscopy with a reconfigurable coded incoherent light-sheet array, Light: Science & Applications (2020). DOI: 10.1038/s41377-020-0245-8
Journal information: Light: Science & Applications
Provided by The University of Hong Kong