March 12, 2020 feature
Heavy metal ion detection and extraction using paper-based atom stamp printed devices
Microfluidic paper-based analytical devices (µPADs) are a promising concept with rapid development in recent years. In a new study published on Nature: Microsystems & Nanoengineering, a team led by Yanfang Guan and Baichuan Sun in electromechanical engineering in China, developed a new technique to engineer µPADs known as atom stamp printing (ASP). The method was cost-effective, easy to operate and allowed high production efficiency with high resolution. As a proof of concept, they used µPADs engineered via the ASP method to detect varied concentrations of copper (Cu2+) via a colorimetric method. The devices achieved a Cu2+ detection limit of 1 mg/L. Guan et al. also created a new paper-based solid-liquid extraction device (PSED) using a three-dimensional (3-D) µPAD with a "3+2" structure and recyclable extraction mode. Due to the characteristics of paper filtration and capillary force, the device could efficiently complete multiple extraction and filtration steps from solid-liquid extraction processes. The PSED platform allowed simple, cost-effective and fast heavy metal ion detection at point-of-care. The work has great promise for applications in food safety and environmental pollution in resource-limited areas.
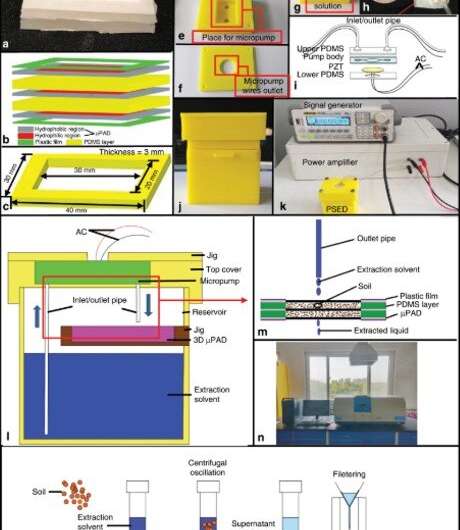
In the 1990s, bioengineers first proposed a conceptual "lab-on-a-chip" (LOC) device based on microfluidic technology. Researchers have since proposed microfluidic paper-based analytical devices (µPADs) to replace traditional microfluidics, including chips based on glass and silicon. Advantages include simple fabrication, low cost, portability and disposability for widespread applications in point-of-care testing. A number of methods were employed to fabricate µPADs including photolithography, wax printing, papercutting and stamping. Atom stamps (AS) or machine-engraved penetrating stamps can be engraved manually, although the practice requires skill and experience. A laser engraving machine can work with common drawing software including AutoCAD and CorelDraw to form a seal stamp that absorbs ink due to its microporous architecture. Guan et al. used the new approach to produce µPADs through AS printing (ASP), where they soaked a stamp of the required pattern in PDMS solvent, printed it on paper and left it in a vacuum drying box to complete fabrication. They then chose the colorimetric method to detect Cu2+.
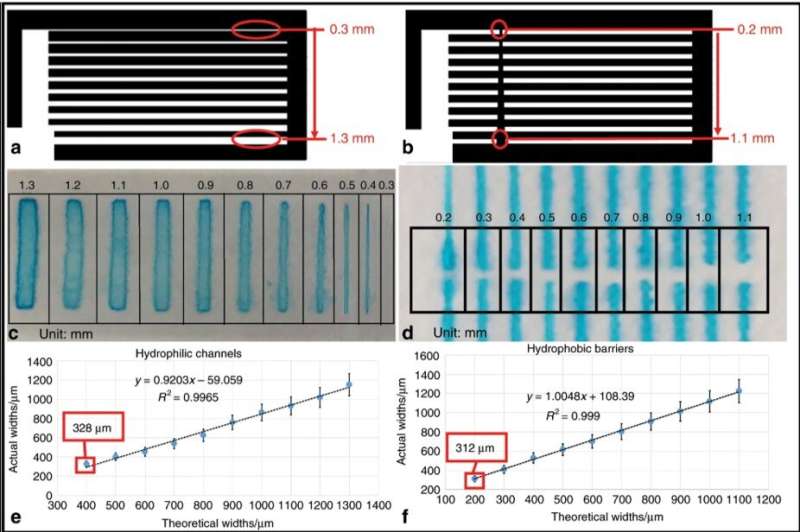
The team demonstrated the versatility of the µPADs by introducing an integrated device for paper-based soil-liquid extraction. The device capitalized on the advantages of the paper including its low cost, portability and filterability to demonstrate superior performance during experimental extraction. They analyzed the resolution of µPADs, as an important metric to regulate their performance, which determined the minimum channel width for fluid flow passages on the paper and for hydrophobic (water-hating) barriers engineered to prevent the flow of fluid. The team observed the flow using a blue dye. The µPADs previously built via laser-cutting provided the highest resolution with a minimum hydrophilic (water-loving) channel width. However, ASP was more efficient compared to preceding techniques used to produce µPADs.
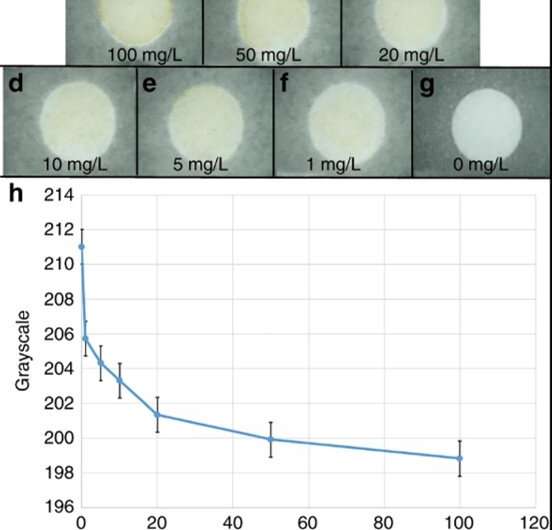
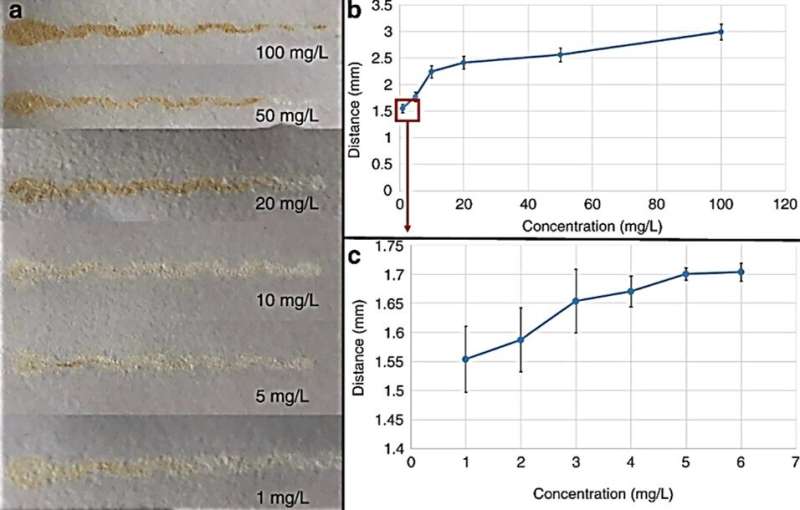
During colorimetric analysis, μPADs with Cu2+ changed from white to yellow, increasing in color with increasing Cu2+ concentration, which Guan et al. quantified using Image J software. The team then determined distance-based detection of Cu2+ solutions as diverse concentrations of solution flowed through the μPAD channels. The length of the yellow band increased with increasing Cu2+ solutions and they observed a constant above 100 mg/L, which they determined as the upper limit of the device. Guan et al. detected a minimum Cu2+ concentration of 1 mg/mL, in accordance to the World Health Organization (WHO) and U.S. Environmental Protection Agency (EPA), where the maximum Cu2+ contamination concentration in drinking water is 2 and 1.3 mg/L, respectively.
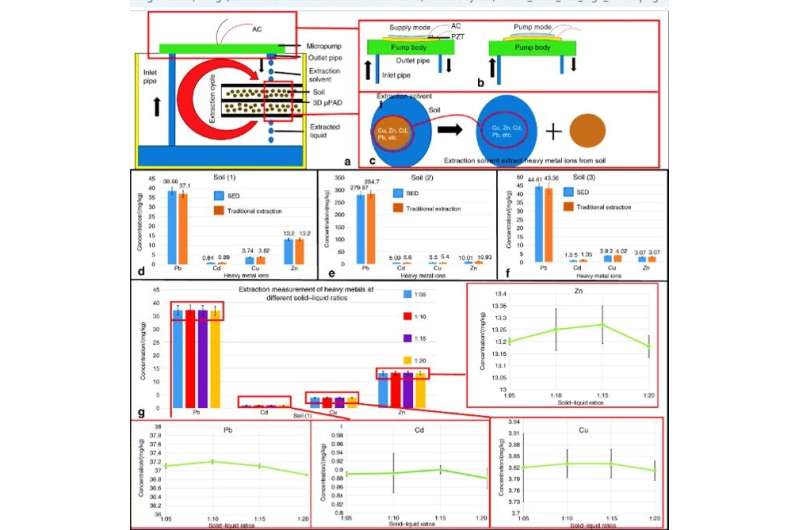
Guan et al. showed the working principle of a paper-based solid-liquid extraction device (PSED), which included a micropump and the microporous character of the filter-paper to complete solid-liquid extraction and filtration. During the process, they stored soil samples on top of the 3-D µPAD and extracted solvent mixed with the soil flowing from the outlet pipe of the micropump. The extraction solvent concomitantly solubilized the heavy metal ions and sucked them through the inlet pipe and pumped them out again in an extraction cycle. Finally, they extracted heavy metal ions including Cu, zinc (Zn), cadmium (Cd) and lead (Pb) from the soil samples via continuous supply and pumping cycles of the micropump.
The concentrations of heavy metal ions obtained from the PSED extraction procedure were similar to traditional methods—proving the effective nature of PSED. The extraction volume required optimization and Guan et al. used more than 30 mL of the extractant as a result. The team optimized the time and found 20 minutes to be sufficient to completely extract heavy metal ions. Each 3-D µPAD could hold 2 g of soil and the entire process of extraction took 40 minutes to complete.
Compared to traditional modes of extraction, the 3-D µPAD technique omitted filtration processes for simpler operation and higher extraction accuracy. The 3-D µPADs are portable, cheap and accessible for simple extraction protocols. The scientists can adjust the size of the device to meet different needs, providing flexibility for real-world applications. In this way, Yanfang Guan and Baichuan Sun developed a new type of detection device known as the µPAD using atomic stamp printing (ASP). They achieved high resolution to form the hydrophilic channels and the hydrophobic barriers of the device. The ASP technique is low cost, has a simple time of activity, allowing short sample preparation for high resolution and higher sensitivity compared to traditional methods.
The ASP fabricated µPADs detected Cu2+ as a proof of concept using a colorimetric method combined with distance-based detection to achieve Cu2+ at a concentration of 1 mg/mL. The team proposed PSED as a new solid-liquid extraction device to extract heavy metal ions from the soil. The device required fewer experimental samples to meet the needs of point-of-care testing, with reduced sample loss. The device maintained high extraction efficiency, low cost and no pollution to meet the demands of solid-liquid extraction. The simple construct can be produced with low cost 3-D printing and is not limited to test soil samples. The team expect to improve the uses of this device to produce high throughput point-of-care testing products.
More information: Yanfang Guan et al. Detection and extraction of heavy metal ions using paper-based analytical devices fabricated via atom stamp printing, Microsystems & Nanoengineering (2020). DOI: 10.1038/s41378-019-0123-9
Eric K. Sackmann et al. The present and future role of microfluidics in biomedical research, Nature (2014). DOI: 10.1038/nature13118
Andres W. Martinez et al. Programmable diagnostic devices made from paper and tape, Lab on a Chip (2010). DOI: 10.1039/c0lc00021c
Journal information: Nature , Lab on a Chip
© 2020 Science X Network