Chemists improve method of extracting natural antioxidants from bay leaves
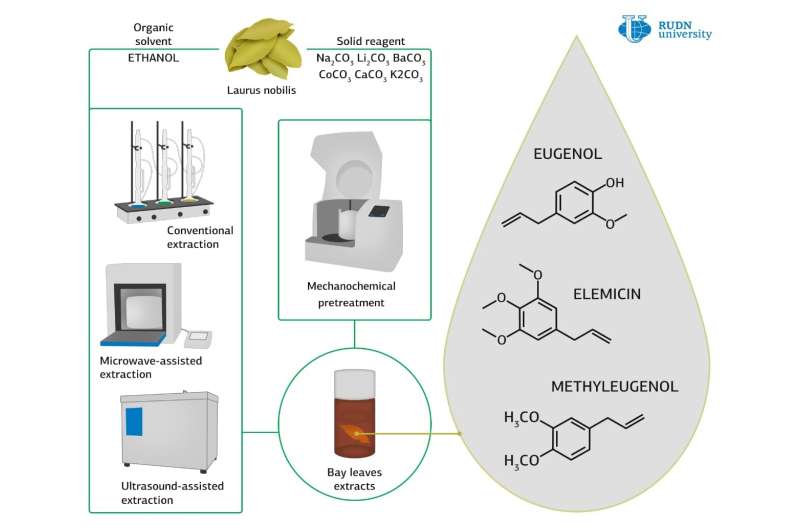
A chemist from RUDN University and his colleagues have proposed using residues from processing bay leaves as raw material for the production of chemically active substances, in particular, antioxidants. The researchers have developed a mechanochemical method for the extraction of antioxidants, which reduces processing time by a factor of 10. The work was published in the journal Industrial Crops and Products.
Isolation of the desired compounds from plant materials by traditional methods takes a lot of time and a large amount of solvent. One promising technology is mechanochemical processing, in which raw materials are processed mechanically to destroy cell walls before extraction. This treatment does not require solvents and reduces production costs. However, there are still very few studies on the use of mechanochemistry for the extraction of natural compounds.
Rafael Luque, director of the scientific center Molecular Design and Synthesis of Innovative Compounds for Medicine, RUDN University, and his colleagues from the University of Cordoba have developed a mechanochemical method that obtains the right compounds from the processing waste of bay leaves.
At the first stage of the experiment, the researchers got the extract from leaves using three solvents: ethanol, methanol and water. Then they used the ultrasonic, microwave, and mechanochemical extraction methods. In the case of the mechanochemical method, the chemists also tested carbonates.
For each of the methods, the authors measured the antioxidant activity of the obtained samples, the total amount of dissolved substances and the amount of phenols. It turned out that the specific antioxidant ability was greater for the extract in ethanol, so in a further study, they used ethanol as the main solvent.
Then the chemists investigated the properties of various carbonates as additives. Lithium carbonate (Li2CO3) was the only one found to be suitable for mechanochemical extraction. The authors then compared mechanochemical processing with microwave and ultrasound processing. In the case of mechanochemical processing, the product yield is higher and the extraction takes less time.
Next, the chemists compared the composition of the extracts obtained by classical, mechanochemical, microwave and ultrasonic extraction. The main component of the extracts is the by-products formed during the degradation of cell walls. The extracts also contained such essential oils as eucalyptol, eugenol, linalool, and caryophyllene. Eucalyptol and linalool have antimicrobial activity, and eugenol and caryophyllene are among the strongest natural antioxidants. Most target substances and the least by-products were extracted by microwave extraction.
The chemists note that mechanochemical extraction is the most promising method for the separation of chemical compounds from plant materials, because with the same content of the necessary substances, the extraction time is reduced by 10 times compared to other technologies. Replacing traditional methods with mechanochemical ones would be a big step forward, meeting all the principles of "green chemistry."
More information: Esther Rincón et al. Mechanochemical extraction of antioxidant phenolic compounds from Mediterranean and medicinal Laurus nobilis: A comparative study with other traditional and green novel techniques, Industrial Crops and Products (2019). DOI: 10.1016/j.indcrop.2019.111805
Provided by RUDN University




















