November 15, 2019 report
A new kind of glue to bond polyethylene
A team of researchers from the University of Victoria and the University of British Columbia, both in Canada, has developed a new kind of glue that is able to bond polyethylene materials together. In their paper published in the journal Science, the group explains how they created a carbene crosslinker with two diazirine motifs. Felix de Zwart, Johan Bootsma and, Bas de Bruin with the University of Amsterdam have published a Perspective piece in the same journal issue outlining the work by the team.
As de Zwart, Bootsma and de Bruin note, scientists would like to develop universal products that could be used for crosslinking polymeric materials. Doing so would allow for creating new products out of such materials as polyethylene and polypropylene—both of which lack crosslinking properties. In this new effort, the team in Canada has developed a crosslinker that can be used to bond such materials together.
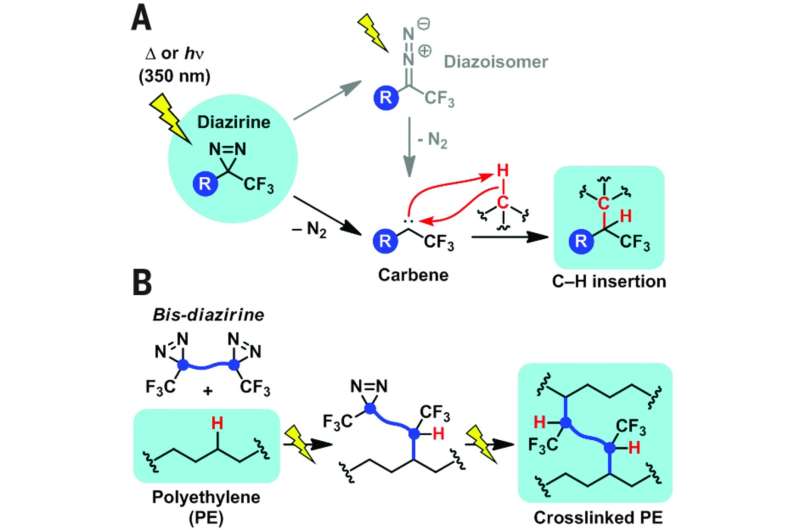
As de Zwart, Bootsma and de Bruin further note, singlet carbenes (and transition metal carbene complexes) are able to undergo direct C-H insertion, resulting in covalent C-C bonds. Carbenes are molecules that have a divalent carbon atom (the carbon atom sits as an intermediary between two other atoms) and are commonly used as catalysts and backbones for polymers. In this new effort, the researchers created a new carbene using an N2-expelling motif that could be used to bond polyethylene materials together.
The researchers report that they "rationally" created a bis-diazirine molecule that decomposes into a carbene under controllable conditions—such as exposure to heat or light. In this instance, "rationally" means that they balanced the risk of explosion or ignition with reactivity—choosing bis-diazirine compounds that offered the best results taking both into consideration. The result was a molecule that was able to insert into C-H polymer bonds. They report that it was capable of crosslinking between assorted polymer chains, including both polyethylene and polypropylene. They note that when used with polypropylene, molecular weight increase was proportional to the amount of crosslinker used. They also looked into the possibility of using their crosslinker with a high-density polyethylene. They note that testing with a commercial product (Super Glue) was ineffective—their product worked much better.
More information: Mathieu L. Lepage et al. A broadly applicable cross-linker for aliphatic polymers containing C–H bonds, Science (2019). DOI: 10.1126/science.aay6230
Journal information: Science
© 2019 Science X Network