Cells control their dance of death
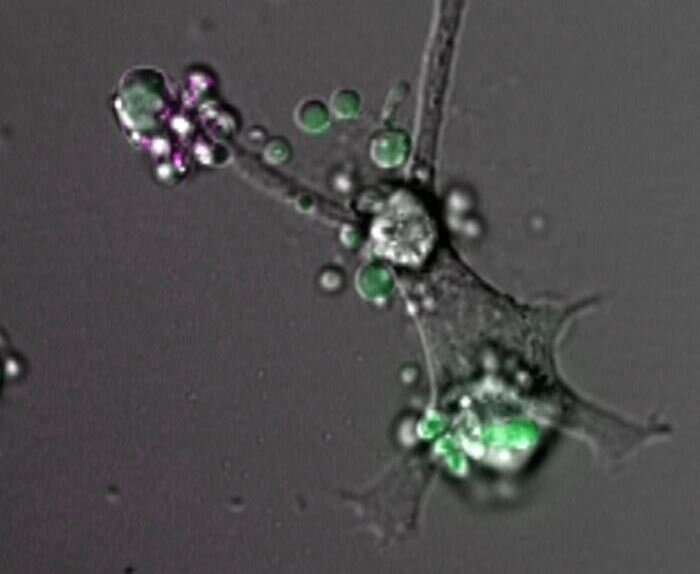
La Trobe University researchers have revealed for the first time how white blood cells control the final moments of their death, helping their own removal from the human body.
Scientists from the La Trobe Institute of Molecular Science (LIMS), working with renowned La Trobe cell biologist Associate Professor Ivan Poon, have identified a protein within dying cells called Plexin B2 that is responsible for coordinating a key stage of apoptosis (cell death).
Published today in Cell Reports, the study is a significant step forward in understanding what may trigger numerous disease states such as inflammation and autoimmunity.
Lead researcher Dr. Georgia Atkin-Smith said the key function of Plexin B2 was discovered when they used CRISPR gene-editing technology to delete the protein from white blood cells.
"Normally, dying white blood cells undergo a dynamic death and shoot out long, beaded, necklace-like structures and fragment into small pieces," Dr. Atkin-Smith said.
"However, since our discovery of this process in 2015, we have not understood how or why this dance of death occurs.
"Our new research identified the protein Plexin B2 as the first molecule which controls this event."
Dr. Atkin-Smith said genetically deleting Plexin B2 prevented dying cells from forming beaded structures.
"Surprisingly, this defect in the cell death process significantly compromised their removal by the garbage trucks of the body, known as phagocytes," Dr. Atkin-Smith said.
"Typically, dying cells send 'find me' and 'eat me' signals to phagocytes, however, deletion of Plexin B2 could limit the number of fragments that are 'ready-to-eat'."
This study follows research—published in June 2019 in Cell Death and Differentiation—from Associate Professor Poon and Dr. Atkin-Smith, led by Dr. Rochelle Tixeira, which also demonstrated the importance of dying cell fragmentation, through a process called blebbing, to aid in the efficient removal of cells by phagocytes.
Dr. Atkin-Smith said understanding how dying cells are removed by phagocytes is fundamental.
"Defects in this process trigger a wide variety of inflammatory disorders like autoimmunity," Dr. Atkin-Smith said.
"Now, for the first time, we have a new insight into what may be the underlining cause of these diseases."
Dr. Atkin-Smith said, collectively, the researchers at Associate Professor Poon's laboratory are world-leaders in the science of dying cell fragmentation.
"Excitingly, the next phase of our research within Associate Professor Poon's laboratory is to take our key basic biology discoveries and examine them in pre-clinical models of autoimmunity," Dr. Atkin-Smith said.
More information: Cell Reports (2019). DOI: 10.1016/j.celrep.2019.10.014
Journal information: Cell Reports
Provided by La Trobe University