December 2, 2019 feature
Cardiac imaging with 3-D cellular resolution using few-mode interferometry to diagnose coronary artery disease
A new imaging technique developed by Biwei Yin and interdisciplinary researchers at the Massachusetts General Hospital and Harvard Medical School in the U.S., provides resolution at the subcellular-level to image the heart's vascular system. As a result, heart researchers can study and diagnose human coronary artery disease with greater precision. Conventionally, cardiologists employ intravascular optical coherence tomography (OCT) to assess the buildup of coronary plaque, which can narrow arteries to cause coronary artery disease.
The OCT technique is, however, limited by a lateral resolution of only 30 microns (µm) therefore researchers are unable to obtain images at the subcellular level to understand the disease. The newly developed few-mode interferometry intravascular imaging system contains a resolution of three microns to provide images of cellular and subcellular structures in the artery wall. The enhanced view can provide detailed information on individual crystals, smooth muscle cells and inflammatory cells with greater precision during disease diagnosis. The research work is now published on Light: Science & Applications.
Optical coherence topography (OCT) is a mainstream imaging method used to obtain cross-sectional reflectance mainly in clinical settings to image a range of human tissues including luminal organs within the body. Intravascular OCT (IVOCT) is of interest to access coronary plaque structure and guide percutaneous coronary intervention (PCI) during coronary artery disease; a leading cause of mortality in the world. Bioengineers and cardiologists have recently demonstrated advanced IVOCT techniques, such as multimodal IVOCT to combine the conventional form with additional imaging and sensing modes, such as fluorescence and near-infra-red spectroscopy. Additional innovations include polarization-sensitive IVOCT to measure tissue birefringence and provide imaging contrast, as well as heartbeat IVOCT to densely image coronary arteries in vivo without introducing motion artefacts. The most challenging technical barrier to increase the lateral resolution of an OCT system includes adjusting the depth of focus (DOF) for cross-sectional imaging. Preceding studies that achieved increased DOF, have a form factor or complexity to prevent intraluminal clinical applications for coronary imaging.
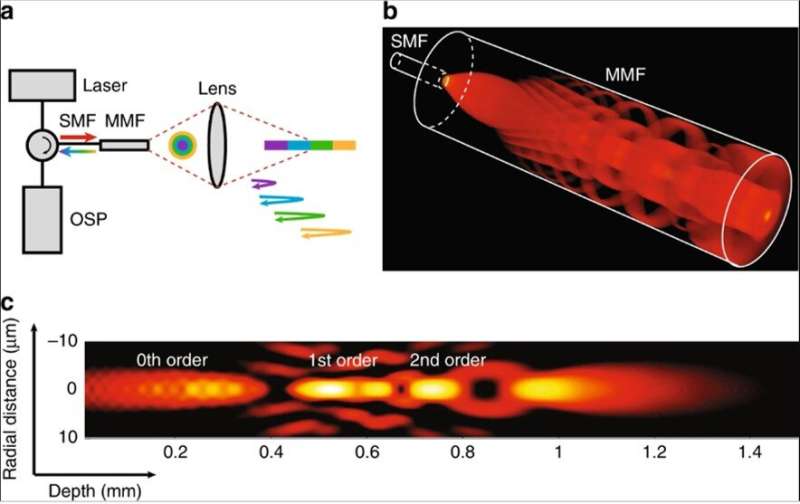
In the present work, Yin et al. described a few-mode interferometry-based intravascular imaging system with extended DOF for cross-sectional imaging at cellular resolution, across a depth range of more than 1 mm. The technology allowed them to observe cellular and sub-cellular structures of intact human-coronary-sized arteries ex vivo and in vivo through a flexible, submillimeter diameter catheter. The researchers used low coherence interferometry that resolved pathlength delay to decode the information carried by each mode travelling at a different optical pathlength in the experimental setup.
Multiple propagation modes could simultaneously interrogate a sample at different depths to transmit the depth-encoded signal through a common channel for processing. The process increased the acquisition capacity of the reflectometry system without additional illumination and detection channels. To visualize the effects, Yin et al. simulated the focused beam field at different depths along the center of the beam path, where scattering particles introduced aberration into the beam field as field disturbance. The self-healing (self-reconstructing) property of the propagation process suggested the independence of each mode in the scattering media.
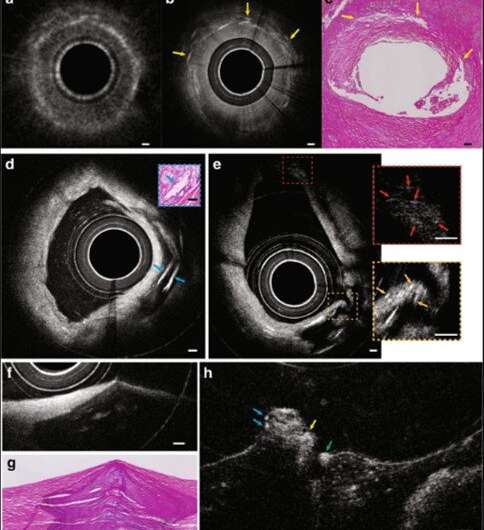
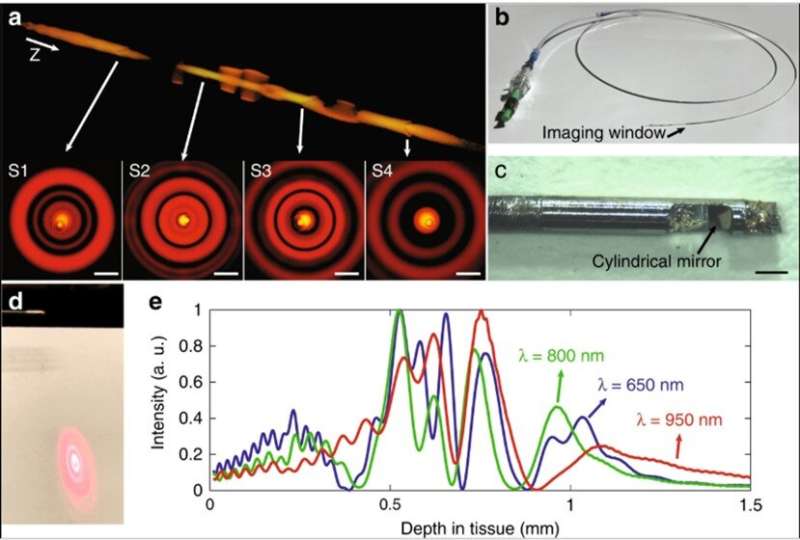
Based on the concept, the research team created an intravascular few-mode interferometry (IVFMI) imaging device with a supercontinuum laser as the light source. They used a low-coherence interferometer as the optical signal processing unit, a catheter for depth encoding and backscattering signal detection, as well as optomechanics for scanning. Using the setup, the researchers conducted a helical scan of the lumen wall for three-dimensional (3-D) reconstruction of the artery. Using a catheter inside an artery they acquired cross-sectional images at 17 frames per second. With an almost 1000-fold improvement in volumetric resolution, the research team resolved cellular and subcellular structures using IVFMI (intravascular few-mode interferometry) in contrast to the conventional IVOCT (Intravascular OCT) method.
For example, when the scientists compared standard IVOCT and IVFMI images corresponding to the same cross-section of a human cadaver coronary artery, they could clearly distinguish densely packed crystals using IVFMI only. In contrast, images obtained using the standard IVOCT technique were blurred and globular, making it more likely to characterize them erroneously as macrophage accumulations. Similarly, the research team observed smooth muscle cells using the IVFMI catheter, which could not be resolved using the conventional IVOCT method.
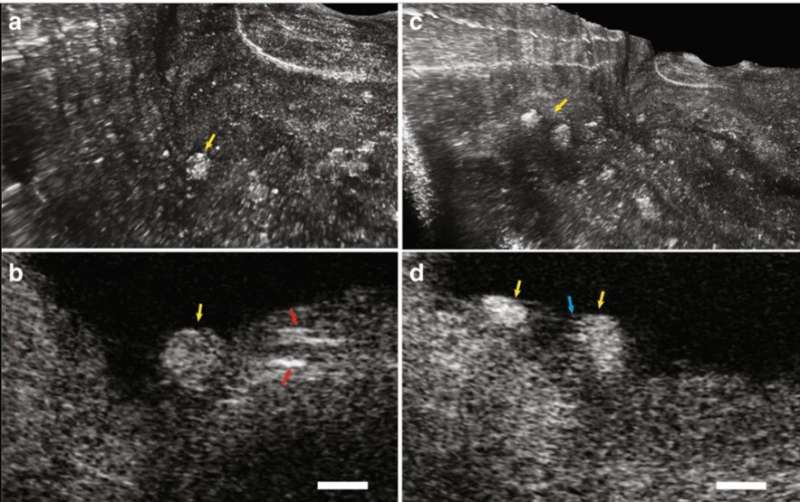
The IVFMI cross-section of an artery also resolved cholesterol crystals that are typically difficult to image using conventional methods due to their notable reflections. As a result of the extended DOF enabled with the IVFMI setup, the researchers resolved microstructures residing a few hundred microns to millimeters away from the catheters sheath, simultaneously in one circumferential scan.
Since inflammatory cells drive the development of atherosclerotic plaques, Yin et al. represented intimal smooth muscle cells and macrophages undergoing diapedesis using IVFMI. The images showed fine detail of the intraluminal mass, including the presence of bright cells such as leucocytes embedded in the fibrin mesh to form what appeared to be a thrombus. The researchers used the IVFMI data obtained from a cadaver coronary lumen wall for 3-D reconstructions and also developed 3-D reconstructions of IVFMI data obtained from a living rabbit aorta with atherosclerotic plaque. They detected the plaque from the normal artery wall by observing the raised surface morphology, which projected into the lumen (inside space of a tubular structure such as an artery).
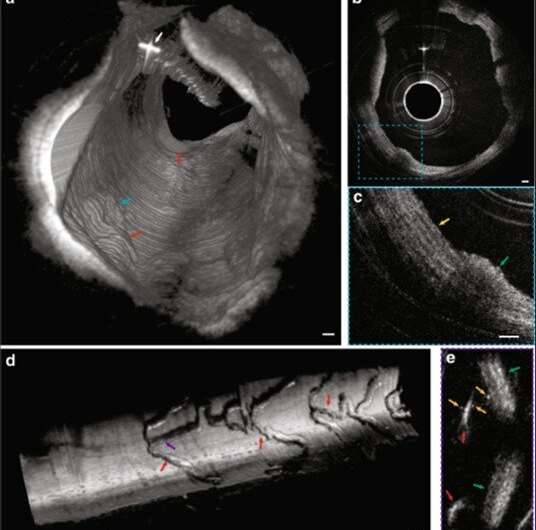
Using cross-sectional images, the team observed a network of collagen and smooth muscle cells in the normal media with improved clarity. They also obtained 3-D reconstructed IVFMI data at a segment of the lumen wall implanted with a stent an hour prior to imaging. The IVFMI process visualized microstructural detail of the stent struts with unprecedented detail for intravascular imaging. Yin et al. observed small, high-reflectivity, micron-sized dots surrounding some of the stent struts and could even identify the fine-detail of microstructural platelets in the images.
In this way, Biwei Yin and colleagues developed and demonstrated a technique to overcome the problems of implementing few-mode interferometry, to increase the depth of focus (DOF) by more than an order of magnitude. The optical configuration technology has a small footprint, depth-encoding capability and transmission stability, with important applications in depth-resolved endomicroscopy. The results confirmed the potential of the new technology to acquire images with a good signal-to-noise ratio and show well-defined disease-relevant cellular and subcellular microstructures within human cadaver coronary arteries ex vivo and rabbit arteries in vivo.
The device is physically and mechanically identical to coronary catheters used for conventional IVOCT imaging in the clinic. These findings indicate the possibility of translating the new IVFMI technique for clinical imaging to view cellular coronary pathology in humans at the cardiac catheterization lab. The technique can be used to view cellular imaging beyond intravascular imaging to include luminal organs such as the gastrointestinal tract and pulmonary tracts to increase clinical diagnostic accuracy.
More information: 1. 3D cellular-resolution imaging in arteries using few-mode interferometry www.nature.com/articles/s41377-019-0211-5 Biwei Yin et al. 21 November 2019, Light: Science & Applications.
2. Simultaneous micromanipulation in multiple planes using a self-reconstructing light beam www.nature.com/articles/nature01007 V. Garcés-Chávez et al. 12 September 2002, Nature.
3. Heartbeat OCT and Motion-Free 3D In Vivo Coronary Artery Microscopy imaging.onlinejacc.org/content/9/5/622.abstract Tianshi Wang et al. 5 May 2016, Journal of American College of Cardiology (JACC): Cardiovascular Imaging.
Journal information: Light: Science & Applications , Nature
© 2019 Science X Network