The structure of master growth regulator
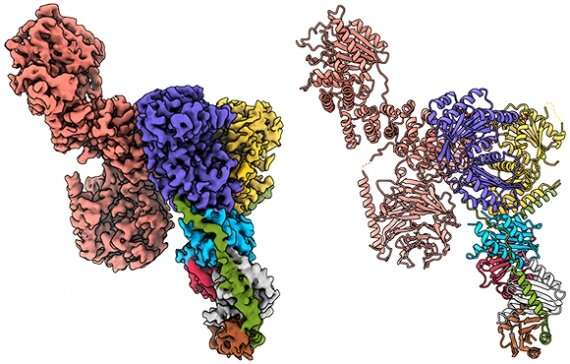
A team of Whitehead Institute scientists has for the first time revealed the molecular structure of a critical growth regulator bound to its partner proteins, creating a fine-grained view of how they interact to sense nutrient levels and control cell growth. Their findings, described in the October 10th online issue of Science, help answer longstanding questions about how the mTORC1 kinase, and its anchoring complex, Rag-Ragulator, work at a molecular level. Using cryo-electron microscopy, the researchers uncover key structures, including a large coiled region and a small, flexible claw. These discoveries help explain the biology of mTORC1 and also lay the foundation for a new generation of drugs that are more precisely tailored to its distinct molecular makeup.
"These interactions are fundamental to the biology of mTORC1, so we and other researchers have been trying to resolve them since the connection of mTORC1 to lysosomes was first discovered in my lab over 10 years ago," says senior author David Sabatini, a Member of Whitehead Institute, a professor of biology at Massachusetts Institute of Technology, and investigator with the Howard Hughes Medical Institute (HHMI). "Now, we have a really deep look at how this important complex works, which opens up a panorama of new research."
mTORC1 is a massive protein complex that enables cells to respond appropriately when food is either abundant or scarce, and has been implicated in a wide range of human diseases, including cancer, diabetes, and neurodegenerative disease. It operates within tiny compartments known as lysosomes—miniature recycling stations of the cell. In order to sense nutrient levels in the lysosome, and become active, mTORC1 must first dock at the lysosomal surface, where it meets up with its anchoring protein (called Rag-Ragulator).
However, this docking is an exquisitely complicated affair. It is regulated by a handful of proteins: an mTORC1 subunit (called Raptor) and the Rag GTPases, which bind Raptor as a non-identical pair and act like a control switch. This switch has four settings: one, which is used when nutrients are high, allows mTORC1 to dock at the lysosome and become active; the other three are used in times of hunger to push the complex away from the lysosomal surface and thereby deactivate it.
"Lacking a detailed structure, there were a lot of unanswered questions about how these proteins work together," says first author Kacper Rogala, a postdoctoral fellow in Sabatini's laboratory. "How does this switch machinery function at the molecular level? How does Raptor know when to bind the Rag GTPases and when not to? We knew we'd need a high-resolution view of the proteins' structure in order to discover the answers."
To achieve that view, Rogala turned to a method known as cryo-electron microscopy or cryo-EM. Instead of creating protein crystals, as in X-ray crystallography, cryo-EM relies on samples that are quickly frozen and then viewed with an electron microscope. But the challenge with mTORC1 and its partner proteins is that they are very dynamic, rapidly coming together and then falling apart, which greatly decreases the odds of capturing an intact complex.
To help turn the tables in their favor, Rogala and his colleagues engineered a variety of single-letter genetic mutations into the Rag GTPases. These mutations were first identified in the tumor DNA of lymphoma patients that exhibited stronger than usual mTORC1 activity. After testing several different mutation combinations, the Whitehead Institute team found the ideal one: two mutations in a single Rag GTPase, which caused the components to linger together in a bound state for slightly longer than usual.
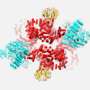
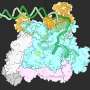
This feat of molecular engineering allowed the researchers to resolve the structure of the Raptor-Rag-Ragulator complex at an extraordinary level of detail—roughly 3 Angstroms, which is about three times the length of a carbon-carbon bond. "At this level of resolution, we can visualize individual amino acids within the proteins and see exactly where their chemical groups are pointing," says Rogala.
With a detailed protein structure in hand, Rogala and his colleagues were able to discern some key structural elements. One, which they describe for the first time, is a claw-like appendage that interacts with one of the Rag GTPases (known as RagC). The other is a large, coiled structure, shaped like a solenoid, that faces RagA.
"We think that, together, these two structures are acting as detectors for the Rag GTPases—so, is the switch in the right configuration for docking at the lysosome or not?" says Rogala.
Researchers at the MRC Laboratory of Molecular Biology in the UK also completed an analysis of these proteins' structures using complementary experimental methods. Rogala and Sabatini collaborated with the group, whose study appears in the same issue of Science.
A deeper understanding of mTORC1 structure is vital not just for understanding how it interacts with its partners. A second, related protein complex (called mTORC2) shares some of the same protein components. Existing drugs against these proteins work non-specifically and often target both mTORC1 and mTORC2 signaling. That lack of specificity can be problematic from a therapeutic perspective—for example, causing unwanted, and often severe, side effects.
"This structure throws open a treasure trove of new biology for us, and that is incredibly exciting," says Sabatini.
More information: Kacper B. Rogala et al. Structural basis for the docking of mTORC1 on the lysosomal surface, Science (2019). DOI: 10.1126/science.aay0166
Architecture of human Rag GTPase heterodimers and their complex with mTORC1. Science. DOI: 10.1126/science.aax3939
Journal information: Science
Provided by Whitehead Institute for Biomedical Research