October 16, 2019 feature
Engineering biomimetic microvascular meshes for subcutaneous islet transplantation
To successfully engineer cell or tissue implants, bioengineers must facilitate their metabolic requirements through vascular regeneration. However, it is challenging to develop a broad strategy for stable and functional vascularization. In a recent report on Nature Communications, Wei Song and colleagues in the interdisciplinary departments of Biological and Environmental Engineering, Medicine, Mechanical and Aerospace Engineering, Clinical Sciences and Bioengineering in the U.S. described highly organized, biomimetic and resilient microvascular meshes. The team engineered them using controllable, anchored self-assembly methods to form microvascular meshes that are almost defect-free and transferrable to diverse substrates, for transplantation.
The scientists promoted the formation of functional blood vessels with a density as high as ~200 vessels per mm-2 within the subcutaneous space of SCID-Beige mice. They demonstrated the possibility of engineering microvascular meshes using human induced pluripotent stem-cell (iPSCs) derived endothelial cells (ECs). The technique opens a way to engineer patient-specific type 1 diabetes treatment by combining microvascular meshes for subcutaneous transplantation of rat islets in SCID-beige mice to achieve correction of chemically induced diabetes for 3 months.
Vasculature is an essential component of any organ or tissue, and vascular regeneration is critical to successfully bioengineer implants. For instance, during cell replacement therapy for type 1 diabetes (T1D), transplanted insulin producing cells rely on the vasculature to function and survive. Bioengineers often use vascular endothelial cells such as human umbilical vein endothelial cells (HUVECs) to spontaneously assemble into tubular structures within the extracellular matrix (ECM). But the resulting structures can be random, uncontrollable and less efficient for microvascular regeneration. Scientists have recently developed three-dimensional (3-D) printing techniques to engineer controlled cellular constructs with embedded vessels. However, it remains challenging to 3-D print resilient and transferrable, high-resolution, microvasculature.
Bioengineering the microvascular meshes
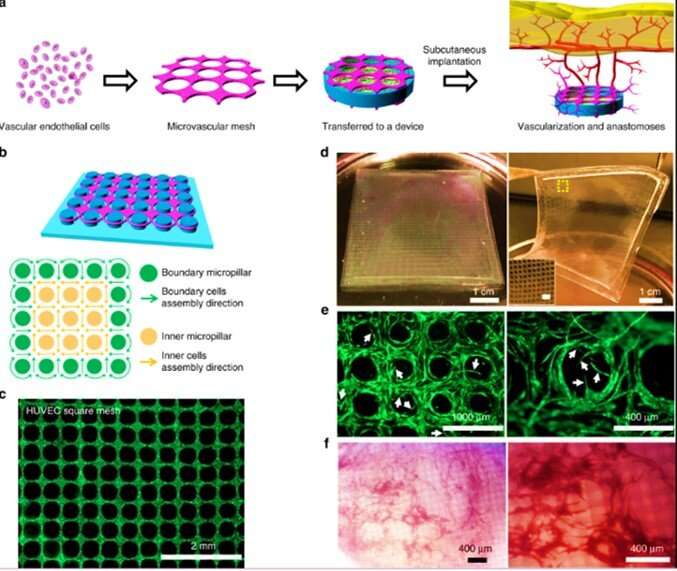
In the present work, Song et al. engineered a microcapillar-based, anchored self-assembly (ASA) strategy to create controllable, transferrable and scalable microvascular meshes for T1D cell treatment therapies. During ASA, the researchers implemented the micropillars to guide self-assembly of endothelial cells within a fibrin matrix. The pillars served as anchoring points to prevent structural shrinkage during cellular maturation for controllable and resilient microvascular meshes.
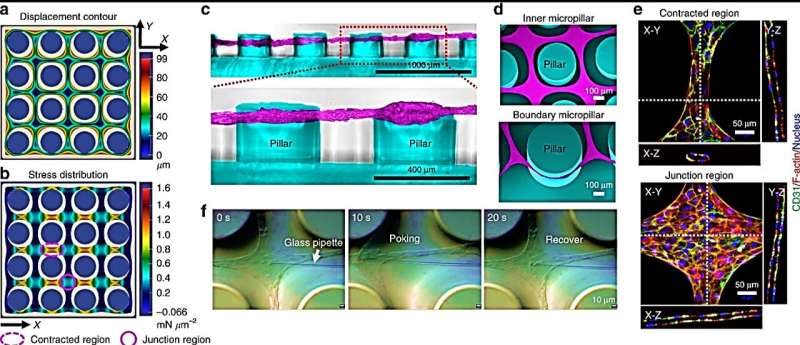
The research team tuned the dimension and arrangement of micropillars to control the geometry of the microvascular mesh. On transplantation, the microvascular meshes promoted the formation of new blood vessels (neovascularization) and vascular anastomoses (connection between two vessels). They developed the meshes using both HUVECs and human induced pluripotent stem cell-derived endothelial cells (iPSC-ECs) suited for patient-specific microvasculature development. Using both types of microvascular meshes, the research team significantly improved vascularization, and corrected diabetes in chemically induced, i.e., streptozotocin (STZ)-based SCID-beige mice for three months. The work provides proof of concept for the use of microvascular meshes in cell replacement therapies for T1D and other diseases.
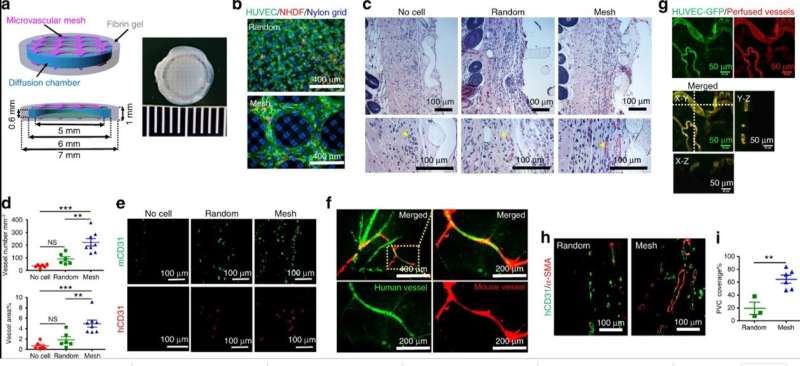
To achieve rapid and functional vascularization around a cellular device, the research team attached a preformed vascular structure with sufficient density and resolution to the device. After transplantation, the structure induced angiogenesis and promoted anastomoses (connection between two vessels) with the host vasculature. During the process, the researchers observed HUVECs together with a fibrin matrix to self-assemble into an almost defect-free square mesh after 2 days of cell culture on a micropillar substrate. They adjusted the size and arrangement of micropillars to precisely control the mesh geometry and dimension. The cells remained viable and stable on micropillar substrates for at least 4 weeks and rapidly generated sprouts on transfer to embed within a fibrin matrix. Due to constant remodeling, the mesh network did not preserve their original shape.
Simulating and experimentally testing the microvascular meshes
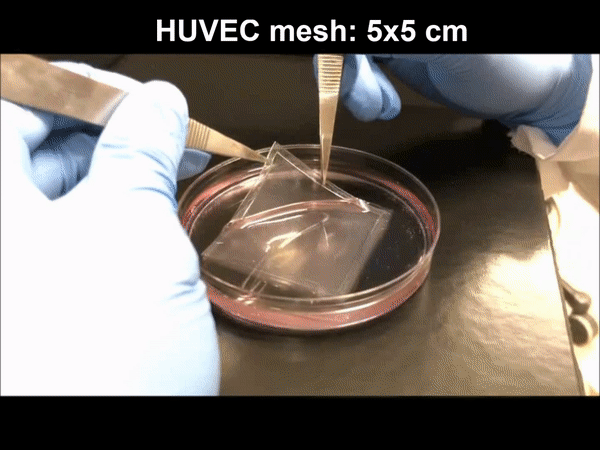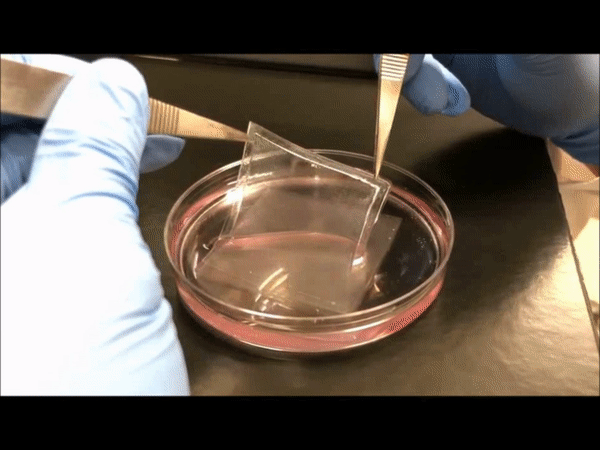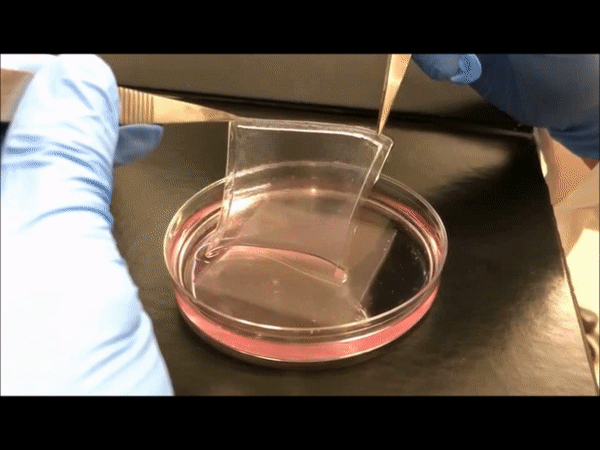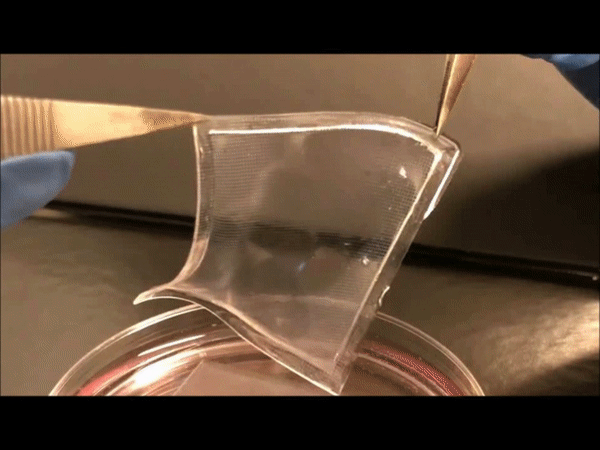
To understand the ASA process, the researchers conducted finite element analysis simulation of the cell assembly process and further tested the microvascular mesh. The simulation considered the contractility of cells on the fibrin matrix to generate an in-plane displacement contour plot of organized mesh structure and stress/strain distribution on the micropillar substrate. The experimental results were consistent with the simulation to validate that microvascular meshes were tightened and suspended between micropillars instead of settling at the bottom of the culture setup. The work supported the hypotheses that stable cell constructs formed by relying on the micropillars. The meshes were also elastic, although mechanically robust and resilient to even withstand poking with a glass pipette. The scientists used these remarkable properties to manipulate and transfer the mesh to diverse substrates without affecting its integrity.
Improved vascularization of micro meshes in SCID-Beige mice.
The research team investigated how microvascular meshes enhanced vascularization by comparing normal HUVEC meshes with normal human dermal fibroblast (NHDFs) added constructs. The combination supported and enhanced vessel formation. The scientists transplanted the constructs within the subcutaneous space of SCID-Beige mice. The subcutaneous space is a poorly vascularized area offering many advantages for cell replacement therapy, including easy access, minimal invasiveness and high transplant capacity. After two weeks, the scientists retrieved the devices and compared vascularization using histology with hematoxylin/eosin (H&E) staining to quantify the blood vessels surrounding the chambers. They revealed a highly vascularized mesh device compared to the control mesh without cells. Immunohistochemical staining further revealed the newly formed vessels to be chimeric in nature, indicating anastomoses and vascular remodeling during vascularization.
To further confirm anastomoses, Song et al. perfused mice with mesh devices via tail injections using two lectin dyes (labeled with green fluorescence protein dye—GFP and a red dye). The overlap of the two different lectin strains confirmed the formation of blood-perfused human vasculatures that anastomosed with the mouse vascular meshes after implanting the HUVEC-GFP/NHDF within the mouse vascular system at different time frames. The results substantiated the ASA-enabled process to transfer microvascular meshes to vascularize cell delivery devices.
Correcting type 1 diabetes (T1D) in SCID-Beige mice using rat islets
Song et al. subsequently investigated if microvascular meshes could improve cell replacement therapy for T1D by loading the rat islets in diffusion chambers and attaching microvascular meshes to them using a fibrin gel mesh device. They then transplanted the constructs subcutaneously in the mouse model with STZ-induced diabetes. After 42 days, the mice showed significantly better blood glucose (BG) control compared to those with control meshes without cells and other random groups. When the research team retrieved the grafts, the BG levels increased in all mice to substantiate the anti-diabetic effects of the devices. The research team performed histological staining of the retrieved devices at day 42 to show significantly increased blood vessels surrounding the islets in the implants from the mesh group; supporting diabetes correction. In total, the results confirmed the effectiveness of transferrable micro-meshes to promote revascularization of donor islets and maintain normoglycemia for up to 3 months in diabetic mice.
Engineering microvascular meshes using human iPSC-ECs.
Song et al. then explored the ASA strategy to other types of ECs (endothelial cells) in a clinical setting using human iPSC-ECs; which offered an unlimited cell source for vascularization with significant clinical potential. The iPSC-ECs behaved similarly to HUVECs in vitro, the research team then implanted the microvascular meshes of diverse geometries within the subcutaneous space of SCID-Beige mice for 2 weeks. They confirmed anastomoses and the formation of blood-perfused iPSC-EC vessels using histology stains.
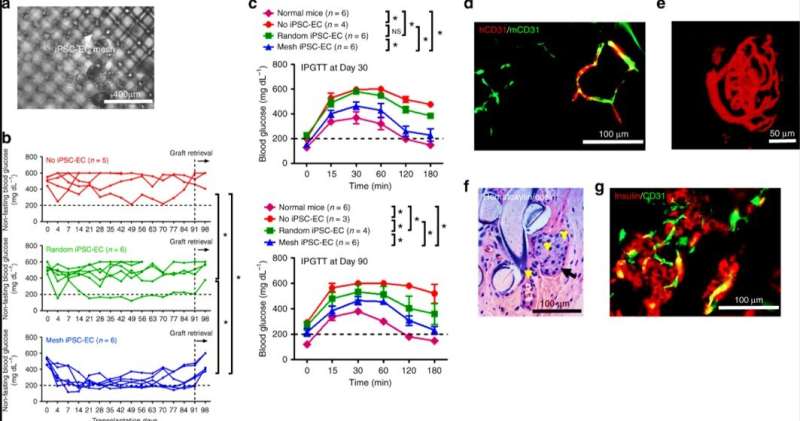
When the scientists similarly attached the iPSC-EC mesh to a diffusion chamber containing rat islets to support the islets. The mesh group led to significantly better diabetes control compared to the control groups in the rat-to-mouse transplantation model. Based on the results, the researchers intend to enhance the function and vascularization of iPSC-EC meshes to improve BG control and glucose responsiveness in transplanted islets. The immunostaining results confirmed the iPSC meshes to promote anastomoses between human and mouse vessels. The results verified the feasibility of engineering patient-specific microvascular meshes during cell replacement therapies for T1D.
In this way, Wei Song and colleagues used spatially arranged micropillars to engineer high-resolution, resilient and transferable microvascular meshes. The micropillar guided the ECs to form patterns with controllable geometry to prevent cells and matrix from shrinking. The biomimetic microvascular mesh was within the same size range of an in vivo capillary bed for enhanced vascularization. The resilient and transferable microvascular meshes allowed researchers to vascularize islets subcutaneously for transplantation within delivery devices during cell replacement therapy in a T1D mouse model. Song et al. expect to expand the ASA as a general approach across other fields of bioengineering to construct geometrically defined, high-resolution live materials at the microscale for cell therapeutics and regenerative medicine.
More information: Wei Song et al. Engineering transferrable microvascular meshes for subcutaneous islet transplantation, Nature Communications (2019). DOI: 10.1038/s41467-019-12373-5
Peter Carmeliet. Angiogenesis in life, disease and medicine, Nature (2005). DOI: 10.1038/nature04478
Boyang Zhang et al. Biodegradable scaffold with built-in vasculature for organ-on-a-chip engineering and direct surgical anastomosis, Nature Materials (2016). DOI: 10.1038/nmat4570
Journal information: Nature , Nature Communications , Nature Materials
© 2019 Science X Network