Sweet! How C. difficile toxin A enters intestinal cells
Clostridiodes difficile infection has become a leading cause of severe, sometimes fatal diarrheal illness. It flourishes best in hospitals and long-term care facilities where people are on long-term antibiotic treatment, but it's also an increasing problem in the community. Much of the damage from C. diff is caused by toxins the bacterium produces, which damage the intestinal lining.
Now, with new research published today in Nature Microbiology, we finally know how C. diff's two primary toxins, A and B, slip into intestinal cells, the first step toward a possible treatment that doesn't involve antibiotics.
"The disease is purely because of the toxins," says Min Dong, Ph.D., who researches bacterial toxins in the Department of Urology at Boston Children's Hospital. "C. difficile strains without toxins can colonize the intestine, but they cause no problem."
Screening for genetic vulnerabilities
In 2016, Dong and his colleagues revealed the entry port for toxin B. Using CRISPR/Cas9 gene editing technology, they screened all genes in human cells for their potential roles in toxin binding and entry into cells. When they mutated the gene for a receptor called Frizzled, the toxin couldn't get into the cells, and intestinal tissues became less sensitive to it.
Using the same approach, the team has now identified the entry port for toxin A. Even better, they found that this toxin's activity can be blocked with molecules already being developed for various medical indications.
The toxin A CRISPR/Cas 9 screens were led Liang Tao, Ph.D., Songhai Tian, Ph.D., and Jie Zhang, Ph.D. in Dong's lab. They found that many genes involved in synthesizing certain sugar molecules were needed for toxin A to bind to cells. These sugars, known as sulfated glycans, are highly abundant on cell surfaces. Viruses, natural growth factors and signaling molecules often use them to enter or communicate with cells.
"When we saw the potential role of sulfated glycans, we sought help from our virology colleagues, Dr. Zhuoming Liu and Dr. Lindsey Robinson-McCarthy at Dr. Sean Whelan's lab at Harvard Medical School," says Dong, the study's senior investigator. "They are familiar with these glycans as attachment factors for various viruses."
A convenient entryway
Together, the researchers established that toxin A uses a broad range of sulfated glycans to bind to cell surfaces. But that wasn't the whole story.
"Just binding to cells is not enough," says Dong. "The toxin has to find a way to enter cells efficiently."
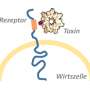
And it has. The top hit from their screen was a gene encoding the low-density lipoprotein receptor (LDLR). This receptor is constantly trafficked, traveling back and forth between the cell surface and the cell interior to bring lipoproteins into cells.
"Toxin A hijacks LDLR to enter cells efficiently," explains Dong. "By combining the initial recognition of glycans with recruitment of LDLR, the toxin maximizes its chance of landing on cell surfaces and can rapidly enter cells."
Biologically speaking, intestinal cells throw out a convenient welcome mat for the toxin. But it's a biology that may be relatively easy to thwart, Dong believes, simply by preventing the toxin from binding to the sugar.
Diversion with a decoy
Commercial interests are already looking to develop sulfated compounds that prevent various molecules from binding to the sulfated glycan receptor. These compounds act as decoys, tying up molecules that would otherwise bind to cells on the intestinal surface.
"The toxin has a preference for sugars on the cell surface," says Dong. "Artificial sugars could block it."
One decoy in development is a sugar-like molecule called GM-1111, similar in structure to the anticoagulant heparin. It was originally designed to suppress the immune response in inflammatory conditions without heparin's anti-coagulation effects, which can lead to bleeding. Its manufacturer, GlycoMira, supplied GM-1111 to Dong's lab, which is repurposing it to counter toxin A.
In a mouse model, GM-1111 reduced toxin-induced fluid accumulation and tissue damage in the colon, an encouraging sign.
The lab's next scientific question to figure out is how toxins A and B work together to contribute to C. diff disease. "Most infections involve both A and B," Dong says. "Now that we have ways to block both toxins, we can try a combination approach."
More information: Sulfated glycosaminoglycans and low-density lipoprotein receptor contribute to Clostridium difficile toxin A entry into cells, Nature Microbiology (2019). DOI: 10.1038/s41564-019-0464-z , www.nature.com/articles/s41564-019-0464-z
Journal information: Nature Microbiology
Provided by Children's Hospital Boston