Changes in pressure, more so than temperature, strongly influence how quickly liquids turn to gas

It's a process so fundamental to everyday life—in everything from your morning coffeemaker to the huge power plant that provides its electricity—that it's often taken for granted: the way a liquid boils away from a hot surface.
Yet surprisingly, this basic process has only now, for the first time, been analyzed in detail at a molecular level, in a new analysis by MIT postdoc Zhengmao Lu, professor of mechanical engineering and department head Evelyn Wang, and three others at MIT and Tokyo University. The study appears in the journal Nature Communications.
"It turns out that for the process of liquid-to-vapor phase change, a fundamental understanding of that is still relatively limited," Wang explains. "While there's been a lot of theories developed, there actually has not been experimental evidence of the fundamental limits of evaporation physics."
It's an important process to understand because it is so ubiquitous. "Evaporation is prevalent in all sorts of different types of systems such as steam generation for power plants, water desalination technologies, membrane distillation, and thermal management, like heat pipes, for example," Wang says. Optimizing the efficiency of such processes requires a clear understanding of the dynamics at play, but in many cases engineers rely on approximations or empirical observations to guide their choices of materials and operating conditions.
By using a new technique to both control and detect temperatures at the surface of an evaporating liquid, the researchers were able to identify a set of universal characteristics involving time, pressure and temperature changes that determine the details of the evaporation process. In the process, they discovered that the key factor determining how fast the liquid could evaporate was not the temperature difference between the surface and the liquid, but rather the difference in pressure between the liquid surface and the ambient vapor.
The "rather simple question" of how a liquid evaporates at a given temperature and pressure, has remained unanswered despite many decades of study, says Pawel Keblinski, professor and head of Department of Materials Science and Engineering at Rensselaer Polytechnic Institute (RPI), who was not involved in this work. "While theorists speculated for over a century, experiment was of little help, as seeing the evaporating liquid-vapor interface and knowing the temperature and pressure near the interfaces is extremely challenging," he says.
This new work, Keblinski says, "brings us closer to the truth." Along with other new observational techniques developed by others, the new insights will "put us on the path to finally quantify the evaporation process after a century of efforts," he says.
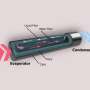
The researchers' success was partly the result of eliminating other factors that complicate the analysis. For example, evaporation of liquid into air is strongly affected by the insulating properties of the air itself, so for these experiments the process was observed in a chamber with only the liquid and vapor present, isolated from the surrounding air. Then, in order to probe the effects right at the boundary between the liquid and the vapor, the researchers used a very thin membrane riddled with small pores to confine the water, heat it up, and measure its temperature.

That membrane, just 200 nanometers (billionths of a meter) thick, made of silicon nitride and coated with gold, carries water through its pores by capillary action, and is electrically heated to cause the water to evaporate. Then, "we also use that membrane as the sensor, to sense the temperature of the evaporating surface in an accurate and noninvasive way," Lu says.
The gold coating of the membrane is crucial, he adds. The electrical resistance of the gold varies directly as a function of the temperature, so by carefully calibrating the system before the experiment, they are able to get a direct reading of the temperature at the exact point where evaporation is taking place, moment by moment, simply by reading the membrane's resistance.
The data they gathered "suggests that the actual driving force or driving potential in this process is not the difference in temperature, but actually the pressure difference," Wang says. "That's what makes everything now aligned to this really nice curve, that matches well with what theory would predict," she says.
While it may sound simple in principle, actually developing the necessary membrane with its 100-nanometer-wide pores, which are made using a method called interference lithography, and getting the whole system to work properly took two years of hard work, she says.
Overall, the findings so far "are consistent with what theory predicts," Lu says, but it is still important to have that confirmation. "While theories have predicted things, there's been no experimental evidence that the theories are correct," Wang adds.
The new findings also provide guidance for engineers designing new evaporation-based systems, providing information on both the selection of the best working fluids for a given situation, as well as the conditions of pressure and removal of ambient air from the system. "Using this system as a guideline you can sort of optimize the working conditions for certain kinds of applications," Lu says.
This team "did a series of elegant experiments designed to confirm theoretical predictions," says Joel Plawsky, professor of chemical and biological engineering at
RPI, who was not involved in this work. "The apparatus was unique and painstakingly difficult to fabricate and operate. The data was exceptional in its quality and detail. Any time one can collapse a large spread of data by developing a dimensionless formulation," that is, one that applies equally well under a wide variety of conditions, "that represents a major advance for engineering," he says.
Plawsly adds, "There are many questions that this work opens up about the behavior of different fluids and of fluid mixtures. One can imagine many years' worth of follow-on work."
More information: Zhengmao Lu et al. A unified relationship for evaporation kinetics at low Mach numbers, Nature Communications (2019). DOI: 10.1038/s41467-019-10209-w
Journal information: Nature Communications
Provided by Massachusetts Institute of Technology
This story is republished courtesy of MIT News (web.mit.edu/newsoffice/), a popular site that covers news about MIT research, innovation and teaching.