Synthetic molecules deliver drugs directly to neuroblastoma cells
Researchers from Universidad Politécnica de Madrid are involved in the development of specific molecular scaffolds which are able to deliver drugs and diagnostic agents to the cells of a neuroblastoma, an aggressive tumor in children.
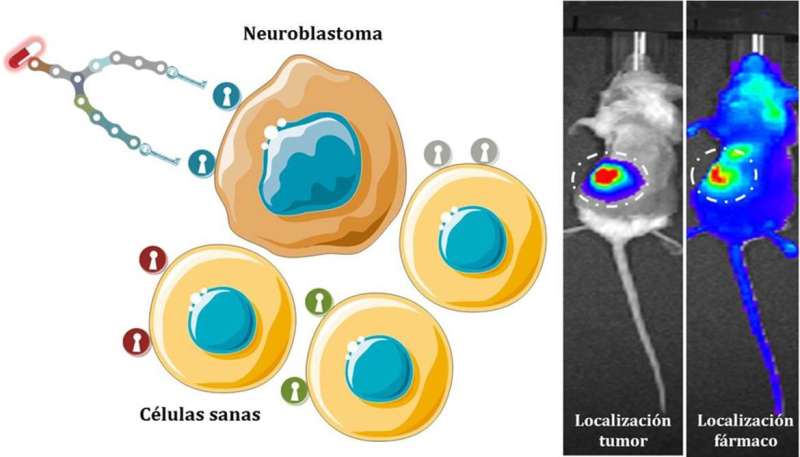
A multidisciplinary team from diverse research centers in Madrid, including various hospitals, Universidad Politécnica de Madrid (UPM) and Complutense (UCM), has developed synthetic molecules which are able to specifically attach to neuroblastoma cells.
The development of a system that is able to recognize cancer cells and specifically deliver drugs to such cells will increase the efficiency of the therapy and reduce the unwanted effects. So far, mice have been used for the trials, but it is expected its application in humans within the next five years.
The neuroblastoma is a very aggressive pediatric tumor with a complex diagnosis when detected in the metastatic state. The current treatments consist of the administration of chemotherapeutic agents in order to kill cancer cells and slow the disease progression.
Unfortunately, these drugs lack specificity against cancer cells and cause numerous side effects and high systemic toxicity, compromising the chances of recovery and quality of life of the patients. In order to find new solutions to improve the treatment, various research centers from Madrid in collaboration with diverse hospitals started a study that is already showing good results.
To this end, researchers have synthesized a family of molecules which are able to specifically bind a protein found in the cell membrane of more than 90% of neuroblastoma cells, the norepinephrine transporter (NET).
"These molecules have a small central structure composed by natural amino acids that work as scaffolds specifically designed to fit in the recognition centers of the NET protein in a similar way that a key fits the lock," Alejandro Baez, a UPM researcher involved in the project, explains. "This way, the process of recognition is highly selective, since the binding only occurs with the suitable 'padlock,' which is only located in the surface of neuroblastoma cells—thus avoiding the healthy cells," Baez concludes.
The effectiveness of these molecular scaffolds was tested in lab mice suffering from this disease and the capacity of transporting both drugs and diagnostic agents were shown. Researchers are currently working on the application of this technology to specific transport of antitumor drugs in neuroblastoma models similar to those that appear in humans.
They are also studying the use of these systems for the improvement of early clinical diagnosis techniques of this disease. "It is expected that the future results will allow us to study its applications in human within two to five years," researchers say.
This project is a long term collaboration of the group of Professor María Vallet (Universidad Complutense de Madrid), Dr. Manuel Ramírez Orellana (Hospital Niño Jesús) and Dr. Alejandro Baeza who is currently a professor at Universidad Politécnica de Madrid.
More information: Gonzalo Villaverde et al. Molecular Scaffolds as Double-Targeting Agents For the Diagnosis and Treatment of Neuroblastoma, Angewandte Chemie International Edition (2018). DOI: 10.1002/anie.201811691
Journal information: Angewandte Chemie International Edition
Provided by Universidad Politécnica de Madrid