May 31, 2019 feature
Quick liquid packaging: Encasing water silhouettes in 3-D polymer membranes for lab-in-a-drop experiments

The ability to confine water in an enclosed compartment without directly manipulating it or using rigid containers is an attractive possibility. In a recent study, Sara Coppola and an interdisciplinary research team in the departments of Biomaterials, Intelligent systems, Industrial Production Engineering and Advanced Biomaterials for Healthcare in Italy, proposed a water-based, bottom-up approach to encase facile, short-lived water silhouettes in a custom-made adaptive suit.
In the work, they used a biocompatible polymer that could self-assemble with unprecedented degrees of freedom on the water surface to produce a thin membrane. They custom designed the polymer film as an external container of a liquid core or as a free-standing layer. The scientists characterized the physical properties and morphology of the membrane and proposed a variety of applications for the phenomenon from the nanoscale to the macroscale. The process could encapsulate cells or microorganisms successfully without harm, opening the way to a breakthrough approach applicable for organ-on-a-chip and lab-in-a-drop experiments. The results are now published in Science Advances.
The possibility of isolating, engineering and shaping materials into 2-D or 3-D objects from the nanometer to the microscale via bottom-up engineering is gaining importance in materials science. Understanding the physics and chemistry of materials will allow a variety of applications in microelectronics, drug delivery, forensics, archeology and paleontology and space research. Materials scientists use a variety of technical methods for microfabrication including two-photon polymerization, soft interference lithography, replica molding and self-folding polymers to shape and isolate the material of interest. However, most materials engineering protocols require chemical and physical pretreatments to gain the desired final properties.
In contrast to the conventional method of using solid molds to create micro- and nanopatterned materials, scientists are now focusing at the air-liquid or liquid-liquid interface to create shells of orderly assembled nanoparticles or crystals to engineer micro- and nanostructured polymeric membranes. The major drawback of the technique is the creation of a polymeric drop immersed in water instead of a free standing polymer suite. In the present work, Coppola et al. started from the existing approach with aims to broaden the experiment to polymer wrap liquid, inorganic and organic micro-objects or microstructured surfaces and remove the liquid core post-fabrication.
The scientists proposed an experimental approach in the present work to directly shape polymer membranes and encapsulate microbodies thereafter. The process consisted of the self-assembly of a biocompatible polymer above the water surface with agility and reproducibility. Coppola et al. chose poly(lactic-co-glycolic acid) (PLGA) due to its tunable structure, drug release efficiency, high biosafety and biocompatibility. They allowed the polymer film to be the external container of a liquid core and proposed using the technique on micropillars, organic and inorganic micro-objects and colloidal particles under mild conditions, to accommodate microorganisms and cells inside the membranes thereafter.
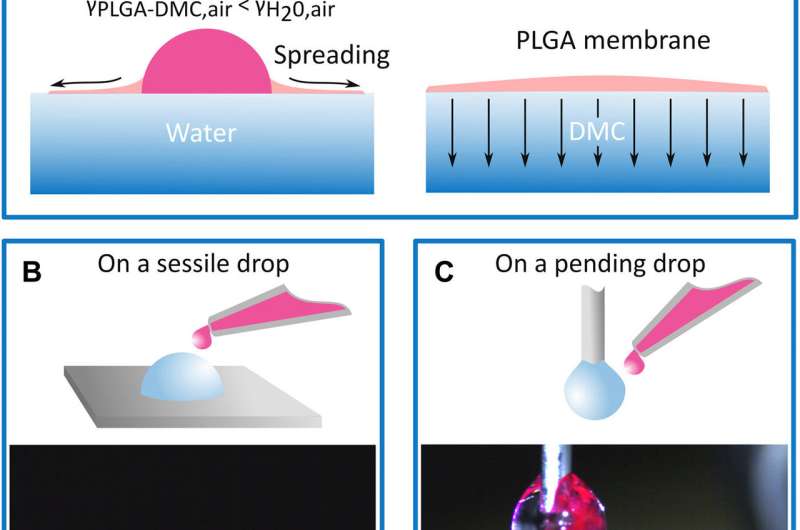
In the experiments, Coppola et al. dissolved a droplet of a biocompatible polymeric solution such as PLGA in dimethyl carbonate (DMC) and placed it on the surface of a water droplet to instantly form a nonporous film. The process allowed the polymer solution to wrap the free water surface, on top of the droplet and create a new interface. The polymer film extended across the free aqueous surface to acquire the shape and structure of the liquid, which was used as a 2-D or 3-D template. They then tested the fabrication process on diverse liquids such as cell culture media, phosphate buffered saline and other buffer solutions containing a water component.
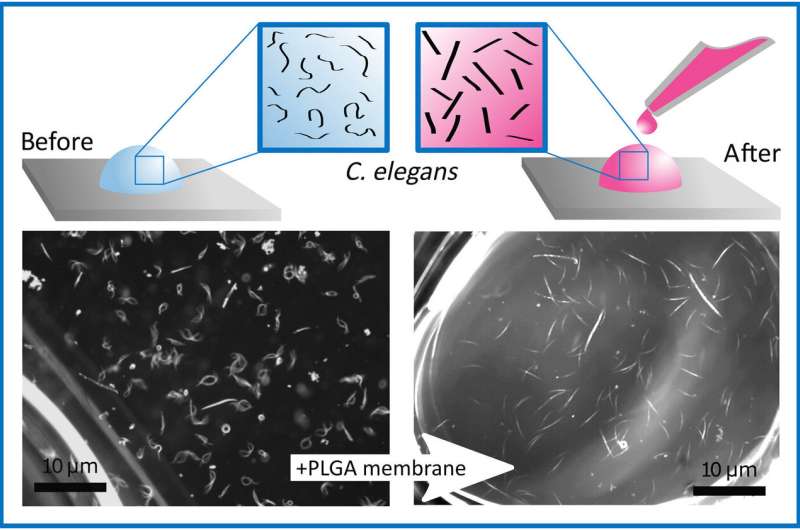
They created a polymeric film even during dynamic and unstable conditions—for instance, on a drop standing on a glass slide and a drop flowing from a needle. To demonstrate total encapsulation of the liquid volume, the scientists formed two separate sessile droplets on the Teflon slide with one encased by the membrane. Upon tilting the surface, the free water droplet moved along the substrate, while the membrane-coated drop remained irremovable and anchored to the glass. In its mechanism of action, the film immediately formed on contact with water and when the solvent evaporated alongside water, the remaining polymer maintained a 3-D structure.
The film did not collapse under atmospheric pressure and the membrane acted as an external coat similar to a polymeric shell on the liquid droplet. The scientists used a variety of membrane characterization methods including scanning electron microscopy (SEM), water contact angle and Young's modulus measurements. The SEM images revealed a nonporous symmetric structure characterized by a homogenous surface and thickness. When they measured the water contact angle on the membrane, the results revealed mild hydrophilicity (water loving) of the polymers. The scientists investigated the mechanical properties of the PLGA membrane and calculated the oxygen permeability and water vapor permeability. The membrane showed a very high permeability to oxygen, which is an important parameter for biomedical applications.

The scientists used the material as an external coating during lab-in-a-drop experiments to form new methods for real-time observations in 3-D. As a proof-of-principle, they studied the behavior of the model organism Caenohabditis elegans in the polymer bubble. For this, they placed the microorganism (MO) in a water solution and wrapped the PLGA membrane around the drop of liquid to show the immediate cessation of MO movement. While C. elegans adhered to the water-PLGA membrane, oxygen flow continued due to membrane permeability for their survival. The abrupt change of MO behavior reversed upon membrane removal to recover usual motility. The process allowed the scientists to observe the MOs without administering harmful drugs to prevent their motion. Coppola et al. propose further experiments to understand the behavior of the organisms within the miniscule polymer drops.
They then tested the possibility of maintaining the phenomenon in the presence of complex contours or obstacles and on hydrogel materials. Using micropillar arrays the scientists observed the polymer membrane to envelope the underlying micropattern and produce peak-and-valley shaped polymer films with arrayed bumps. Such functionalities will allow Coppola et al. to design cell culturing substrates, scaffolds for tissue engineering and drug delivery systems using the polymer systems.
Similarly, when they tested the technique with hydrogel materials by simply dispensing a polymer drop or spraying the polymer above a rotating hydrogel cylinder, they were able to form a continuous polymer film. Using the method, they produced polymer films with different molds in the shape of microcubes, rhombus and cylinders for a variety of applications.
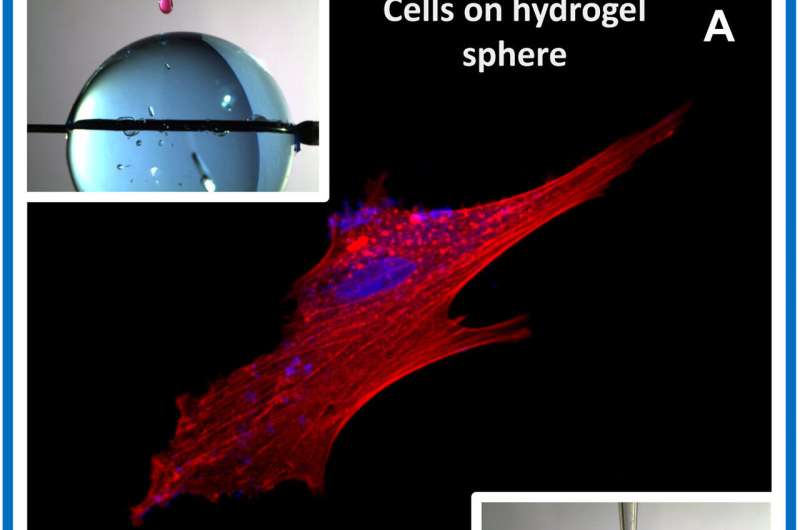
The scientists used the polymer-hydrogel constructs as a scaffold for cell culture experiments to observe cell growth on various shapes including microsphere cubes and polymer patterns. After 24 hours of culturing human mesenchymal stem cells (hMSCs) in PLGA, the scientists visualized the cytoskeleton and nuclei to show cell body elongation on the polymer film; indicating adequate cell adherence. The proposed technique did not harm cell cultures or microorganisms to form a new and simple method to engineer polymer films with potential scalability for microfluidic organ-on-chips.
In this way, Coppola et al. developed an environment-friendly, cost-effective and water-based bottom-up engineering approach to allow a biopolymer to self-assemble on a droplet of water and on other 3-D templates. The scientists propose using the materials for a range of applications in biomedicine for wound healing, as lab-in-a-drop and on lab-on-a-chip devices. They envision optimized functionalities of the polymeric film with semiconductor nanoparticles or quantum dots to open new routes in clinical phototherapy in living systems in the future.
More information: Sara Coppola et al. Quick liquid packaging: Encasing water silhouettes by three-dimensional polymer membranes, Science Advances (2019). DOI: 10.1126/sciadv.aat5189
D. H. Gracias. Forming Electrical Networks in Three Dimensions by Self-Assembly, Science (2002). DOI: 10.1126/science.289.5482.1170
Christopher Moraes et al. Defined topologically-complex protein matrices to manipulate cell shape via three-dimensional fiber-like patterns, Lab Chip (2014). DOI: 10.1039/c4lc00122b
Journal information: Science Advances , Science
© 2019 Science X Network