May 1, 2019 report
Why a drop of oil bounces in a water/ethanol gradient and eventually falls to the bottom of a jar

A team of researchers working at the University of Twente has solved the mystery of why a drop of oil bounces repeatedly when dropped in a water/ethanol gradient but eventually falls to the bottom of a jar. In their paper published in the journal Physical Review Letters, the group describes their study of the odd behavior.
In physics, the Marangoni effect describes differences in surface tension between liquids that can lead to seemingly strange behaviors such as "tears" forming in a glass of wine with a lot of alcohol in it. In this new effort, the researchers explored the Marangoni effect as it applies to the bouncing of oil drops in a jar containing a water/ethanol gradient.
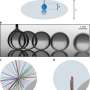
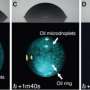
To set up the experiment, the researchers carefully poured ethanol into a jar of water, avoiding too much mixing, such that a density gradient formed naturally. Then, they introduced a single drop of oil, in this case, anethole, into the container. The drop sunk slowly through the mixture before rapidly rebounding straight back up. The drop does this several times before eventually sinking all the way to the bottom of the container. After careful study of the whole scenario, the researchers discovered what was actually happening.
At the onset, the drop falls due to gravity—the researchers found that as it does so, the surface tension on the drop is different on the top and bottom (the Marangoni effect), resulting in an energy buildup. Once it reaches a tipping point, the energy is released, pushing the drop back up toward the surface. But each time it falls and rebounds, the amount of stress that builds up lessens, likely due to the mixing of the water and ethanol as the drop moves down and up repeatedly. Eventually, the amount of energy released is not sufficient to propel the drop upward, and it falls to the bottom of the container. The researchers note that the same scenario can be replicated using other types of oil and liquid density gradients.
More information: Yanshen Li et al. Bouncing Oil Droplet in a Stratified Liquid and its Sudden Death, Physical Review Letters (2019). DOI: 10.1103/PhysRevLett.122.154502 . On Arxiv: arxiv.org/abs/1812.01498
Journal information: Physical Review Letters
© 2019 Science X Network