May 3, 2019 report
A closer look at the molecular mechanism that switches control of activation of eIF2 by eIF2B

A team of researchers at the RIKEN Center for Biosystems Dynamics Research in Japan has developed a tool to prevent neurogenerative diseases. They have demonstrated a way to observe the molecular mechanism that switches control of activation of eIF2 by eIF2B when a cell undergoes stress. In their paper published in the journal Science, the group describes their use of cryo-electron microscopy to better understand what happens to cells that are exposed to stress.
Cells continuously undergo a process known as translation in which ribosomes in cytoplasm synthesize proteins after transcription of DNA to RNA. But sometimes, this process is interrupted by an external event. Such events are known as stress—exposure to ultraviolet light is one common example. The researchers note that translation uses a lot of energy, thus it makes sense for cells to shut it down when a stress event occurs—it saves energy and reduces the chances of errors in the proteins that are synthesized.
Prior research has shown that when a cell "senses" stress, the translational initiation factor eIF2 is phosphorylated. Under normal circumstances, eIF2 is activated by eIF2B, another translational initiation factor. But when the cell is under stress and eIF2 is phosphorylated, the function of eIF2B is inhibited, preventing translation. Scientists have been working to understand the molecular mechanism involved in discontinuation of translation due to stress, but the means of activation of eIF2 by eIF2B is still unknown. In this new effort, the researchers have found a way to look inside the cell nucleus to see what actually happens as syntheses of proteins ceases during stress.
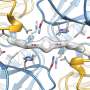
The researchers observed the structure of both eIF2 and eIF2B using cryo-electron microscopy. Doing so showed that the orientation of eIF2 as it was bound to eIF2B differed greatly depending on whether eIF2 had been phosphorylated. They also found that eIF2B had a two-fold symmetric structure and that the phosphorylation of eIF2 could be considered a mechanism that not only prevented the activation of phosphorylated eIF2, but also prevented the activation of other translational initiation factors.
More information: Kazuhiro Kashiwagi et al. Structural basis for eIF2B inhibition in integrated stress response, Science (2019). DOI: 10.1126/science.aaw4104
Journal information: Science
© 2019 Science X Network