Assessing battery performance: Compared to what?

Scientists must often ask themselves, compared to what? How do the results we generate in the laboratory compare with those obtained by others? How do our theoretical calculations compare with experimental data?
Answering such questions is especially crucial for researchers and developers of lithium-ion batteries. Invented four decades ago, lithium-ion batteries now power most portable electronics like laptops and power tools. They are also being developed to meet the high energy storage demands for powering electric vehicles and electric grids. New designs with different compositions of electrode and electrolyte—the two key battery components—are constantly coming online.
Assessing whether an innovation in electrode or electrolyte material is actually an improvement requires comparing it to other test results. However, there is no "one size fits all" standard for battery testing. Methods for testing batteries can vary widely.
Argonne battery researcher Ira Bloom notes, "Industrial engineers and researchers from governmental and academic labs often devise their own procedures for characterizing lithium-ion batteries based on the battery technology's intended application. This makes the comparison of any technological innovations extremely complicated."
A team from the U.S. Department of Energy's (DOE) Argonne National Laboratory, University of Warwick, OVO Energy, Hawaii National Energy Institute, and Jaguar Land Rover has reviewed the literature on the various methods used around the world to characterize the performance of lithium-ion batteries in order to provide insight on best practices.
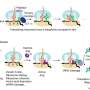
Typically, battery researchers use three parameters to define electrochemical performance: capacity, open-circuit voltage, and resistance. Capacity is a measure of the total charge stored in a battery. The open-circuit voltage is the voltage available from a battery with no current flow. It represents the battery's maximum voltage. The resistance is the degree to which the component materials impede the flow of electric current, resulting in a voltage drop.
The problem is that, depending on battery application, researchers may measure these parameters under different test conditions (temperature, rate of discharge, state of charge, etc.), and thereby obtain a different battery operating life. Battery resistance, for example, can be measured with either a direct current or alternating current.
"It's complicated," observes Anup Barai, a principal investigator and senior research fellow at the University of Warwick. "The appropriateness of a test depends on what the investigator is studying. Our review provides guidance on the most appropriate test method for a given situation." To that end, the team has produced an easy-to-use table comparing eight test methods, including the main equipment needed, the information generated, and the advantages and drawbacks for each.
"Our hope," Bloom adds, "is that our results may one day lead to more reliably comparable methods for testing lithium-ion batteries tailored to different applications."
The study, entitled "A comparison of methodologies for the non-invasive characterisation of commercial Li-ion cells," recently appeared in the online version of the journal Progress in Energy and Combustion Science.
More information: Anup Barai et al, A comparison of methodologies for the non-invasive characterisation of commercial Li-ion cells, Progress in Energy and Combustion Science (2019). DOI: 10.1016/j.pecs.2019.01.001
Provided by Argonne National Laboratory