May 20, 2019 report
A way to determine the absolute stereochemistry of small, organic molecules
A team of researchers from several institutions in Czech Republic has developed a way to determine the absolute stereochemistry (3-D spatial configuration) of small, organic molecules. In their paper published in the journal Science, the group describes their new technique and how well it worked. Hongyi Xu and Xiaodong Zou with Stockholm University, have published a Perspective piece on the work done by the team in the same journal issue.
As the researchers note, the current method for determining the absolute configuration of molecules that have chiral centers is done via X-ray crystallography. The measurement is based on observing how X-rays fired at molecules bounce around. Unfortunately, this method only works on relatively large crystal structures. Efforts to use a similar technique on smaller crystals based on electron diffraction have fallen short of expectations due to the fragile nature of the target—nanocrystals are destroyed by the energy in the electron beams. In this new effort, the researchers have found a way to overcome this problem, which allowed them to determine the stereochemistry of very small crystals for the first time. This is a pretty big deal, Xu and Zou note, because the U.S. FDA and the European Medicines Agency require absolute configuration information for a prospective new drug before it can be approved. This requirement has held back the creation and sale of drugs based on nanocrystals, as pharmaceutical companies had no way to fulfill the requirement.
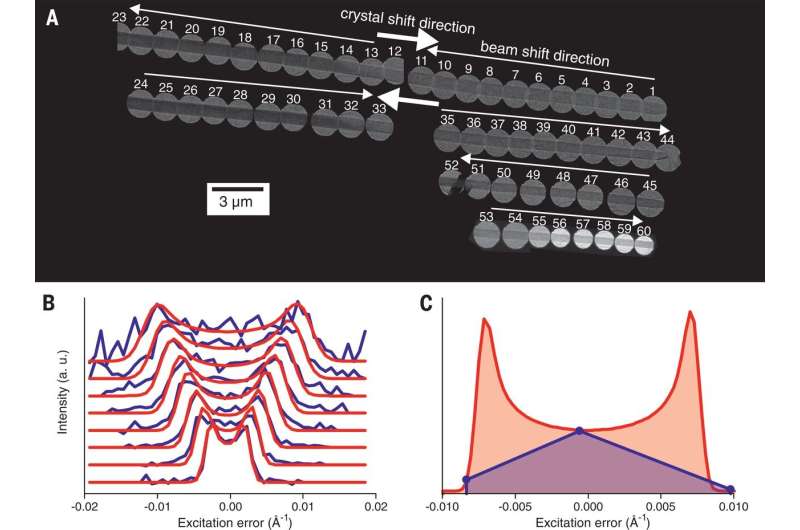
To overcome the problem of electron beams destroying nanocrystals before their stereochemistry could be recorded, the researchers simply used more beams—four of them. They fired them all at once at different parts of the nanocrystal and recorded information regarding the diffracting that occurred before the nanocrystal was destroyed.
Xu and Zou note that X-rays scatter only once when used to determine the configuration of a molecule—with electron diffraction, electrons scatter multiple times, and as they do so, the intensities of their diffractions change—sensors that read such changes are able to measure such dynamical diffraction effects. The result was a description of the absolute stereochemistry of a given molecule. Xu and Zou suggest that the new technique is likely to open the door to development of new materials used in drug design.
More information: Petr Brázda et al. Electron diffraction determines molecular absolute configuration in a pharmaceutical nanocrystal, Science (2019). DOI: 10.1126/science.aaw2560
Journal information: Science
© 2019 Science X Network