Molecules that curb errant proteins of AL amyloidosis point to new type of therapy
Scientists at Scripps Research have identified a group of small molecules that prevent structural changes to proteins that are at the root of AL amyloidosis, a progressive and often fatal disease.
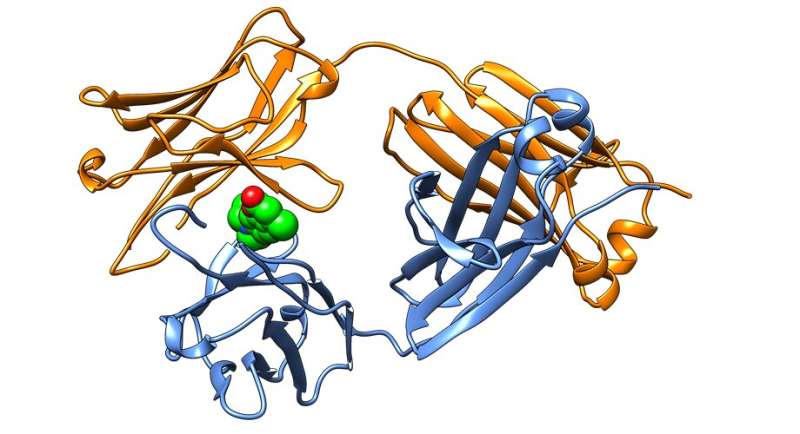
The small molecules bind to immunoglobulin proteins, which play an essential role in the body's immune system, and then stabilize subunits of the protein called immunoglobulin light chains. Binding and stabilizing the light chains in their native shape prevents them from misfolding and forming the toxic plaques found in patients with AL amyloidosis.
By labeling light chains with fluorophores and coupling shape changes to cleavage by proteinase K, nearly a million small molecules were screened for their ability to prevent the disease-associated structural changes using fluorescence polarization. Using this strategy as the basis for a high-throughput screen and various distinct counter-screens to eliminate artifacts, the team discovered multiple small-molecule drug candidates that prevented immunoglobulin light chains from misfolding and aggregating in a test tube. The approach, if successful in humans, could change the course of disease.
"Because we have identified the immunoglobulin light chain small molecule binding site conferring stabilization by crystallography, we believe these findings represent a blueprint for making a much-needed drug for AL amyloidosis," says Scripps Research chemistry professor Jeffery Kelly, Ph.D., who led the research. "If we can block the aggregation of newly secreted light chains and prevent them from being degraded into aggregation-prone fragments, we hope to slow or even halt disease progression, especially for those patients exhibiting cardiac involvement."
Amyloidosis is a rare and likely underdiagnosed disease that takes many forms. AL amyloidosis, also called light chain amyloidosis, is the second most common systemic amyloid disease, with some 4,500 new cases every year in the U.S.
The plasma cells of people with AL amyloidosis produce a component of antibodies known as immunoglobulin light chains, often instead of antibodies, as a result of a plasma cell cancer. The light chains misfold and/or clump together into small fibers that are toxic to organs. Over time, these so-called amyloid deposits progressively interfere with healthy function of the heart, kidneys, liver and other parts of the body.
Scripps Research's Gareth Morgan, Ph.D., lead author of the study, emphasizes that AL patients with organ involvement, particularly cardiac involvement, are often too sick to tolerate chemotherapy. The hope is that immunoglobulin light chain kinetic stabilizers will be able to serve as a first treatment for these patients so that they can ultimately tolerate chemotherapy.
The Scripps Research team's findings appear in the Proceedings of the National Academy of Sciences. In addition to describing their kinetic stabilization approach for treating AL amyloidosis, the researchers say they envision a straightforward method for identifying the patients who would be most likely to benefit from the treatment.
Kelly says the drug mechanism that the team identified is analogous to a different drug that also originated at Scripps Research: Tafamidis, which stabilizes the protein transthyretin to treat the most common systemic amyloid disease that affects the heart or other organs. Kelly invented tafamidis along with Evan Powers, Ph.D., to treat transthyretin amyloidoses. Now owned by Pfizer, tafamidis is approved in Europe under the name Vyndaqel and was recently submitted to the FDA for approval in the U.S.
Nicholas Yan, a graduate student and co-first author on the study, is now developing more potent and selective immunoglobulin light chain kinetic stabilizers with drug-like properties to be used in humans, studies being carried out in collaboration with Reji Nair, Ph.D., a post-doctoral fellow, also in collaboration with others at Scripps Research.
More information: Gareth J. Morgan et al, Stabilization of amyloidogenic immunoglobulin light chains by small molecules, Proceedings of the National Academy of Sciences (2019). DOI: 10.1073/pnas.1817567116
Journal information: Proceedings of the National Academy of Sciences
Provided by The Scripps Research Institute