In cellular world, strong relationships the key to healing old wounds
Yale scientists with colleagues at University College London have taken the next step towards unravelling how cells work together during wound closure, a question that could be fundamental to determining optimal healing rates after injury or disease.
How cells behave, the rate at which they repair skin tissue, and how they coordinate the production of mechanical energy, has been poorly understood by researchers.
Yale scientists have now identified new processes of cell movement in wound healing in outer layer, or "epithelial" tissue abrasions. The findings are published in the journal Nature Physics.
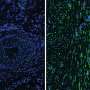
Using lasers to eliminate individual epithelial cells, the researchers "tracked" the movement of remaining cells atop fluorescent hydrogels to calculate the rate of wound deformation and closure, and the total mechanical energy force being generated.
In general, cells move by using cytoskeletal muscle proteins or "lamellipodia" at the leading edge of the cell to propel the whole structure across a substrate.
However, the researchers found that, in the case of smaller abrasions, cells at the wound edge act collectively using two different mechanisms to close a wound: the more common lamellipodium, and also a "purse-string"—a thick bundle across the leading edge of the wound that contracts the rim of cells inwards, closing the wound.
No matter which mechanism was chosen, or where a combination was deployed, the cells maintained a constant rate of energy and closure speed.
"Our results provide a robust model of how cells collectively coordinate their behaviors to dynamically regulate tissue-scale mechanical outputs," said Michael Murrell, assistant professor of biomedical engineering and physics and senior author of the article. "This universal behavior determines how fast your wounds heal."
The researchers aim to expand their work to examine the fundamental laws that determine the rate at which similar biological processes happen, particularly in relation to the consumption of energy by cancer cells.
More information: Visar Ajeti et al. Wound healing coordinates actin architectures to regulate mechanical work, Nature Physics (2019). DOI: 10.1038/s41567-019-0485-9
Journal information: Nature Physics
Provided by Yale University