Unique interface and unexpected behavior help explain how heavy metals act
Refining platinum, plutonium, or certain other metals often depends on how the metal behaves at liquid interfaces. The challenge? Scientists have limited ways to analyze the details of liquid interfaces. Now, researchers described in significant detail how water molecules surround a platinum-based ion. Their description includes an unexpectedly complex structure that forms on the liquid's surface. The team noticed no less than three different forms of water wrapping around the ion. The complex water structure is unexpected and it creates unusual behaviors compared to those observed around smaller and lighter ions under similar conditions.
Refining platinum and other precious metals involves moving the desired metal from one liquid to another. But little is known about how that transfer works on the molecular scale. This work helps explain how platinum and other heavy metals move and react across liquids.
Solvent extraction, the go-to technology for reprocessing nuclear waste or refining rare earths and precious metals, involves the preferential transfer of a targeted chemical species between two immiscible phases through a liquid-liquid interface. Relatively little molecular-scale information is known about the mechanism or mechanisms of this transfer across an interface. Why? In large part, the knowledge gap is because most experimental probes cannot interrogate liquid interfaces directly. Even the best surface sensitive probes are only sensitive to limited components of interfacial structure.
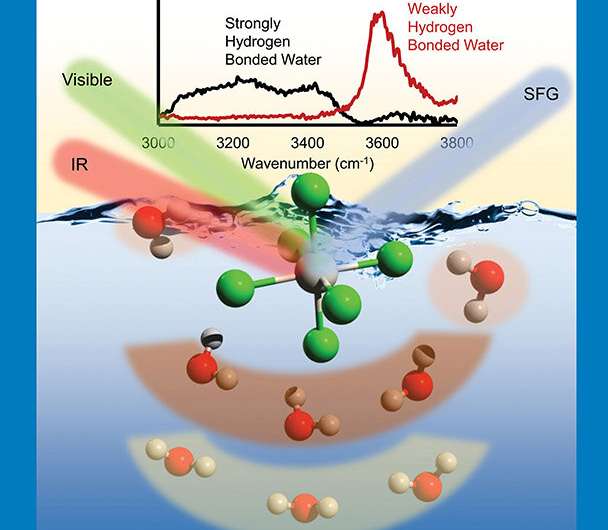
To provide a molecular-scale description of heavy, anionic-complexes, such as PtCl62-, with positively charged extractant molecules at the air/water interface, researchers combined synchrotron X-ray experiments, surface-sensitive sum-frequency generation (SFG) spectroscopy, and molecular dynamics simulations. X-rays provided by the Advanced Photon Source probed structural features of surface-located metal ions. A combination of scattering and fluorescence measurements uncovered a two-step adsorption process that depends on the metal concentration in solution. SFG spectroscopy revealed a unique water structure related to this adsorption. A novel sub-ensemble analysis of molecular dynamics simulations further clarified details of the interfacial water structure resulting from the strongly hydrated anionic complexes. The research showed that after adsorption, the PtCl62- complexes partially retain their first and second hydration spheres. It is possible to identify three types of water molecules around the ions, differentiated by their orientation and hydrogen bonding motifs. The results have major implications for predictive modeling of charged-ions at air/water interfaces.
More information: William Rock et al. Heavy Anionic Complex Creates a Unique Water Structure at a Soft Charged Interface, The Journal of Physical Chemistry C (2018). DOI: 10.1021/acs.jpcc.8b08419
Ahmet Uysal et al. Two-Step Adsorption of PtCl62– Complexes at a Charged Langmuir Monolayer: Role of Hydration and Ion Correlations, The Journal of Physical Chemistry C (2017). DOI: 10.1021/acs.jpcc.7b09350
Journal information: Journal of Physical Chemistry C
Provided by US Department of Energy