Researchers achieve solid state thermochemiluminescence with crystals
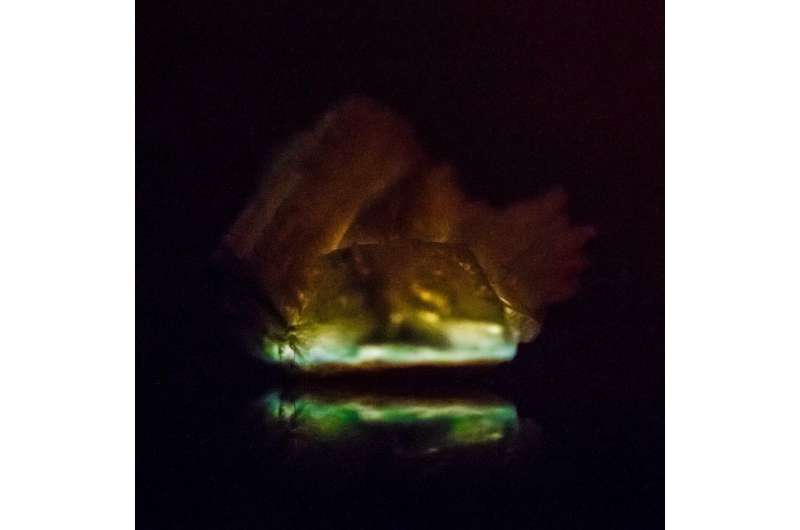
The findings, published in the journal Nature Communications, demonstrate that this fundamental process of transduction of energy—heat, applied to a material, generates light—can be achieved in pure solid materials of millimeter or centimeter size.
In general, chemiluminescence reactions are only known to be possible in solution (a process that, for example, allows glow sticks to give off light) where typically two chemicals easily react, resulting in the emission of visible light. That, however, was not reported in bulk solids.
To explore whether this process would also work in the pure solid state, researchers prepared several organic peroxides and grew centimeter-size crystals—a challenging task, because these compounds are typically known to be very unstable. When heat was applied to the crystals, the molecules decomposed and generated light that ranged from red to blue to green, depending on the chemical composition.
"Our findings open up an unexplored direction in chemiluminescence research with possible applications in both materials research and life science," said lead researcher Dr. Stefan Schramm, a post-doctoral associate in NYU Abu Dhabi's Naumov research group. "We provide fundamentally new insights into this form of energy transduction, which has the potential to be used in developing new solar energy harvesting technologies or for monitoring of initiators in the polymer industry."
NYU Abu Dhabi Associate Professor of Chemistry and the study's supervisor Dr. Panče Naumov said: "This first observation of direct conversion of heat into light in a bulk material, a work that was conducted over more than two years, required building of a new, very sensitive microscope and other special instrumentation. Not only does it demonstrates the fundamental principles of energy transduction that are central to both chemistry and physics, but it also sets the basis for applications in optoelectronics and sensing technology."
More information: Nature Communications (2019). DOI: 10.1038/s41467-019-08816-8
Journal information: Nature Communications
Provided by New York University