Luminescent bacteria in squid light organ drive systemic changes in host
A new study published in Proceedings of the National Academy of Sciences by researchers at the University of Hawai'i (UH) at Mānoa School of Ocean and Earth Science and Technology (SOEST), revealed that luminescent bacteria, which live harmoniously inside the Hawaiian bobtail squid's light organ, change the gene expression in other organs of their squid host.
Bacteria have been driving animal biology since the origin of animals, and most animal-bacteria associations benefit the lives of their host organism in a relationship called symbiosis. Understanding the interdependence of microbes and other organisms, including humans, is the frontier of medicine and environmental health.
The Hawaiian bobtail squid is active at night in the shallow waters of the Hawaiian archipelago. It emits from its body the light produced by its bacterial partner to camouflage against moonlight and starlight. The animal modulates the bioluminescence output of its microbes in response to variations in environmental light, such as attenuation with cloud cover. This behavior is believed to be controlled through feedback between the eye and light organ.
For nearly three decades, the laboratories of director Margaret McFall-Ngai and researcher Edward Ruby at the Pacific Biosciences Research Center (PBRC) in SOEST, have used the Hawaiian bobtail squid and its light-producing symbiont, Vibrio fischeri, as a model biological system to unravel the details of this mysterious and vital relationship.
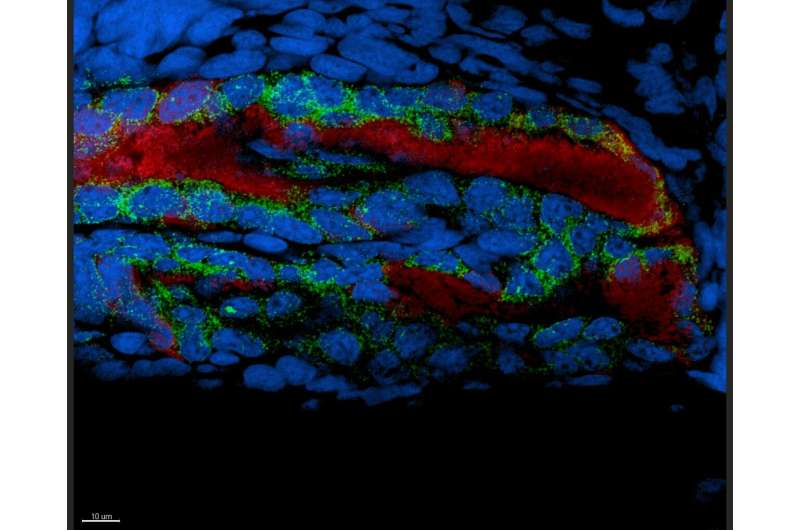
Silvia Moriano-Gutierrez, graduate student in PBRC with McFall-Ngai, asked the question: does Vibrio colonization of the Hawaiian bobtail squid light organ influence gene expression in other parts of the squid's body?
To investigate this question, Moriano-Gutierrez used two different strains of the symbiont—the wild type, which is brightly luminescent, and one that doesn't make light, the 'delta lux strain.' Under three conditions, the team assessed which squid-host genes are expressed in the light organ itself, and in the eye, which is hypothesized to work with the light organ to coordinate host light output, and the gill, which is a primary immune organ in the squid: squid not colonized with Vibrio, those colonized by the wild-type bacteria, and those colonized by the delta lux strain.
"My first surprise was that, in comparing the early influences of colonization by either wild-type bacteria or light-defective ones, we found that the most significant driver of gene expression in the light organ is NOT the presence of the bacteria themselves, but rather the presence of bacteria that make light," said Moriano-Gutierrez. "In other words, light production by the symbionts was a more significant driver of host gene expression."
The fact that the gene expression responds more to light production than to bacterial presence underscores the importance to the animal of the bacteria doing their job of producing bioluminesence. Interestingly, within 48 hours, a delta-lux strain is subsequently rejected by the host, correlating the transcriptional response of the light organ with its ability to sanction a dark strain.
Next, the researchers assessed how the varying conditions affected tissues remote from the symbionts, that is, the eye and gill. Both of these organs responded to these conditions with their own unique gene expression signature. The gills responded to the presence of both wild-type and dark mutants similarly.
"However, the next big surprise was that, while the eye responded with robust changes in the expression of dozens of genes when the light organ is colonized by wild-type, when colonized by the dark mutant, NO genes changed their regulation in the eye," said Ruby. "The data support the idea that coordination between eye and light organ is important for the use of light in the animal's behavior."
"In this way, the onset of symbiosis orchestrates a coordinated, regulated expression of multiple genes, across different organs, both colonized and distant, and may be triggered by diverse symbiont signals, indicating that the dialog between host and symbionts is a more complex conversation than we anticipated," said McFall-Ngai.
With future research, the team will address which other possible features of the symbiont trigger such responses in host tissues, and how those signals get to remote tissues, in an effort to understand the mechanism underlying the complexity of host-microbe communication.
Diverse microbiomes present a challenge for deciphering the signaling between a varied microbial community and host tissues. By using the squid-vibrio system, in which Vibrio fischeri is the only bacterium that can establish a symbiotic association in the light organ, we reduce this complexity to only two partners. In that way, it becomes more manageable to identify and interpret the dialog within the association, revealing strategies that might be relevant for understanding more complex systems like the human gut microbiome. A Hawaiian species is leading the way as a model system for understanding how microorganisms benefit the lives of their hosts.
More information: Silvia Moriano-Gutierrez el al., "Critical symbiont signals drive both local and systemic changes in diel and developmental host gene expression," PNAS (2019). www.pnas.org/cgi/doi/10.1073/pnas.1819897116
Journal information: Proceedings of the National Academy of Sciences
Provided by University of Hawaii at Manoa