Elucidating cellular responses to force
Accumulated evidence suggests that physical force plays an important role in developmental processes of fertilized animal eggs. During embryogenesis, a variety of cell populations actively migrate and change their positions, generating various types of force (e.g., traction force, compression force) that influence the properties of surrounding tissues. This in turn enables normal development in which tissue arrangement is highly orchestrated. However, how embryonic cells and tissues respond to these forces remains poorly understood.
Tackling this important question was made possible by an international collaboration between two laboratories with expertise in proteomics and developmental biology. Professor Ileana Cristea from Princeton University (USA) and Professor Naoto Ueno from the National Institute for Basic Biology (Japan), along with their respective research groups, attempted to answer this question. Together, they characterized global phosphorylation and protein abundance intracellular changes in embryos to which physical force was applied.
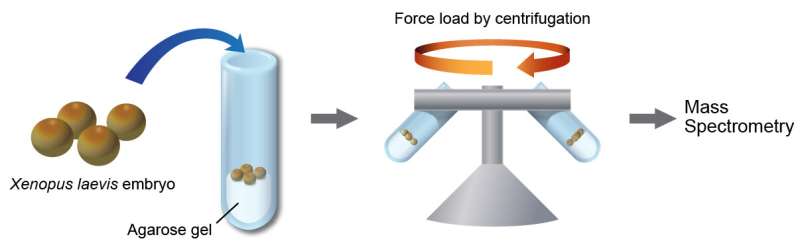
"In this study, we applied force by centrifuging Xenopus embryos, and we extensively analyzed the changes in the phosphorylation state of proteins in them. Since Xenopus eggs are relatively larger than other organisms' eggs, we were able to obtain sufficient amounts of protein for the analysis from a small number of embryos." said Dr. Noriyuki Kinoshita, a member of the research team.
Using a sensitive and quantitative mass spectrometry-based approach, the research team was able to identify phosphorylations on over 9,000 peptides (i.e., fragments of proteins).
Prof. Cristea highlights the novelty and expected impact of this research by indicating that "This is the first study of global temporal alterations in protein phosphorylation in response to mechanical force in any biological system. We integrated methods that allowed us to globally characterize the dynamic phosphoproteome with targeted approaches that we designed to monitor the temporality and relative abundance of specific phosphorylation events with great accuracy. We further placed this knowledge of phosphorylation-mediated signaling in the context of protein abundance changes. Overall, this allowed us to discover precise modulatory points in response to force. Additionally, given the contribution of mechanical forces to both tissue homeostasis and progression of different diseases, we hope that our methods and findings will provide a valuable platform for future investigations in mechanobiology and signal transduction."

The first author of the paper, Dr. Hashimoto said, "It was interesting to note that a variety of protein kinases, such as PAK2 and PKC, in the embryo are activated by force stimulation for only 10 minutes. It was also surprising to find that proteins constituting focal adhesion and tight junctions were among those most prominently phosphorylated. This work also unveiled a previously unrecognized crosstalk between the FAK pathway and the PKA and PKC pathway. In addition, we have been able to demonstrate that centrifugation reinforces tight junctions, leading to the epithelialization of the tissue."
"In particular, it was intriguing to find that upon force stimulation, ZO-1 (one of the tight junction components) is accumulated to tight junctions to strengthen its structure, which is a characterictic of mesenchymal-epithelial transition (MET)-like change. This phenomenon is the opposite to epithelial-mesenchymal transition (EMT) found in some cells during development, wound healing, and cancer invasion/metastasis. We speculate that in embryos, there must be a feedback mechanism that enhances the robustness of tissues to resist deformations induced by force." Prof. Ueno said.
These results were published on March 6th, 2019 in the journal Cell Systems.
More information: Yutaka Hashimoto et al, Mechanical Force Induces Phosphorylation-Mediated Signaling that Underlies Tissue Response and Robustness in Xenopus Embryos, Cell Systems (2019). DOI: 10.1016/j.cels.2019.01.006
Journal information: Cell Systems
Provided by National Institutes of Natural Sciences