Scientists develop metal-free photocatalyst to purify pathogen-rich water in minutes
Scientists around the world have been wracking their brains to solve the global problem of clean water scarcity. Chinese scientists now report a new way to purify pathogen-rich water with high efficiency and without using metal.
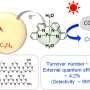
With a metal-free photocatalyst jointly developed by researchers from Yangzhou University (YZU) and the Institute of Process Engineering (IPE) of the Chinese Academy of Sciences, this method can provide standard drinking water in 30 minutes with disinfection efficiency over 99.9999 percent under visible light irradiation. This discovery was published in Chem on January 24.
Photocatalytic disinfection is a promising approach due to its low cost and direct use of sunlight. However, current popular metal-based nanomaterial catalysts may lead to secondary pollution due to the inevitable release of metal ions. Metal-free photocatalysts developed later, however, have much lower efficiency.
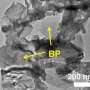
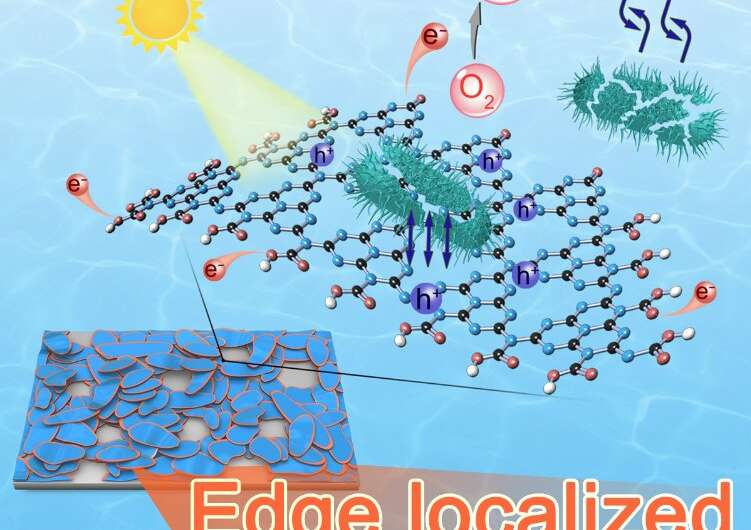
"We investigated a facile wet-chemical method to site-selectively introduce certain groups at the edges of graphitic carbon nitride (g-C3N4) nanosheets," said WANG Dan, a principal investigator at IPE.
These introduced groups not only induced an upward bending of their energy band, improving oxygen molecule adsorption, but also promoted the separation of photo-generated electron-hole pairs, according to WANG.
"Its first-order disinfection rate was five times higher than that of previously reported best metal-free photocatalysts with only 1/10 catalyst consumption," said WANG Chengyin from YZU. The catalytic activity was also comparable to the best metal-based photocatalyst.
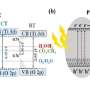
Moreover, this photocatalyst can be fixed on substrates, thus significantly reducing the potential ecotoxicity caused by dispersed nanomaterials in drinkable water.
A facile protocol for incorporating this metal-free photocatalyst into portable devices for highly efficient water disinfection is now available, according to researchers.
"This work presents a facile preparation strategy for highly efficient metal-free photo-disinfection catalysts," said WANG Dan. "It also provides a new strategy for controlling the surface charge densities of carbon materials."
Journal information: Chem
Provided by Chinese Academy of Sciences