New molecular tool identifies sugar-protein attachments
Researchers at Johns Hopkins Medicine have developed a new molecular tool they call EXoO, which decodes where on proteins specific sugars are attached—a possible modification due to disease. The study, published in issue 14 of Molecular Systems Biology, describes the development of the tool and its successful use on human blood, tumors and immune cells.
Half of all proteins made in human cells have sugar molecules attached to them, the most common of which are N-glycans and O-glycans. Until now, O-glycans have been more difficult to study because there were not adequate tools to identify them. And, whereas proteins are coded and made according to the DNA blueprint, whether and how many sugars are attached to proteins can vary, especially in disease conditions.
"The biggest challenge in the field of glycobiology has been to pin down what sugars are conjugated on where and on which proteins, and we've now developed a reliable way to do that," says Weiming Yang, Ph.D., a research associate in pathology at the Johns Hopkins University School of Medicine. "Moreover, we have shown that EXoO can be used in all types of samples including tissues, body fluids and cells."
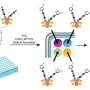
The team developed EXoO by combining different reactions and taking advantage of a process the team had developed for studying other sugar-linked proteins and a bacterial enzyme called OpeRATOR known to cut proteins at the attachment sites of O-glycans. In short, protein samples are first digested into smaller pieces, then those pieces are attached to a solid support, which is treated with the enzyme OpeRATOR that releases small pieces of proteins at the O-glycan attachment sites. Those bits of proteins then are analyzed to determine where the sugar is attached.
"This was great. We showed that EXoO is the first tool that can both identify the site and define the site-specific glycan," says Yang.
To establish that the new process works, the team first used EXoO on a well-studied glycol-fetal calf protein, known to contain six potential O-glycan attachment sites. After running this protein through EXoO, the team was able to confirm all six known sites as well as identify a seventh site.
The team then asked if EXoO could be used on a larger scale with complex mixtures of proteins. So it applied the EXoO method to normal and cancerous kidney tissue samples from three patients with clear cell renal carcinoma, T cells and blood serum. From the kidney tissues, the researchers were able to map 35,848 protein pieces to 2,804 O-glycan-containing proteins with 1,781 attachment sites from 592 proteins; from the T cells, 4,623 protein pieces to 1,982 O-glycan proteins with 1,295 attachment sites from 590 proteins; and from blood serum, 6,157 protein pieces to 1,060 O-glycan proteins with 732 attachment sites from 306 total proteins.
Upon comparing its data to work done by others previously and collected in three glycoproteome databases, the research team's results revealed 2,580 O-glycan sites that had never before been reported, an increase of 94 percent of known sites.
The researchers found 56 proteins that were different in O-glycan attachment when comparing normal to tumor kidney tissues, whereas two well-known proteins not associated with kidney cancer showed new changes in tumor cells compared to normal cells. The researchers say these results suggest that O-glycan attachments to proteins is dynamic and can be very specific to disease.
"We are hopeful that this tool will be useful to researchers and those studying the roles of O-linked glycosylation in normal biology and diseases," says Hui Zhang, Ph.D., M.S., a professor of pathology at the Johns Hopkins University School of Medicine and senior author on the report.
Journal information: Molecular Systems Biology
Provided by Johns Hopkins University School of Medicine