Channels for the supply of energy
Working in cooperation with international colleagues, researchers from the University of Freiburg have described how water-insoluble membrane proteins are transported through the aqueous space between the mitochondrial membranes with the aid of chaperone proteins. The membrane proteins enable the cellular powerhouses to import and export small biomolecules. Thus the team led by Prof. Dr. Nils Wiedemann from Freiburg and Dr. Paul Schanda from Grenoble (France) together with researchers from the University of Copenhagen (Denmark) and the University of Tübingen has answered a fundamental question about the formation of mitochondria. The European Research Council (ERC) funded the research with both a Consolidator Grant and a Starting Grant. The scientists have published their results in the science journal Cell.
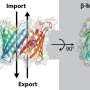
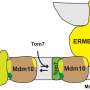
In the same way that the human body consists of various organs, eukaryotic cells contain small organelles such as the mitochondria, which synthesize the energy molecule adenosine triphosphate (ATP). The total amount of ATP that the mitochondrial membranes transport to supply the cells each day is roughly as much as the individual's body weight. This process depends on special channel and transporter protein molecules that are present in the inner membrane and outer membrane of mitochondria. These channels and transporters are produced outside the mitochondria and are transported across the outer membrane. Although these protein molecules are not soluble in water, they have to be transported through the aqueous intermembrane space, so that they can be integrated into the outer or inner mitochondrial membrane.
To achieve this, the intermembrane space contains special TIM chaperone proteins, which bind the channel and transporter proteins to facilitate their transport through the intermembrane space. To identify the molecular mechanism of this process Dr. Katharina Weinhäupl performed structural work and Caroline Lindau performed functional mitochondrial studies which complemented each other. The results show that the ring-shaped TIM chaperones have six water-repellent brackets to which the channels and transporters are loosely attached to prevent their aggregation. This is important because many diseases such as Alzheimer's or Parkinson's are associated with the formation of aggregates of protein molecules. Likewise, a malfunction of the TIM chaperones can cause Mohr-Tranebjærg syndrome with neurological deafness and movement disorders.
More information: Katharina Weinhäupl et al, Structural Basis of Membrane Protein Chaperoning through the Mitochondrial Intermembrane Space, Cell (2018). DOI: 10.1016/j.cell.2018.10.039
Journal information: Cell
Provided by University of Freiburg