New method for hydroboration of alkynes: Radicals induce unusual selectivity
A combination of organoboron and radical chemistry generates unusual trans-selectivity in hydroboration of alkynes. The use of N-heterocyclic carbene boranes is key to the success of this chemical transformation. This study is expected to open the door to the development of new boron-containing materials.
Organoboron compounds have been widely used to create new organic molecules since Prof. Suzuki, a 2010 Nobel Prize winner in chemistry, developed palladium-catalyzed cross coupling reactions with organoboron compounds (Suzuki coupling). In addition, many boron-containing compounds themselves are promising materials. They can be used as medicines and organic electro-luminescence materials because of boron's unique electronic nature.
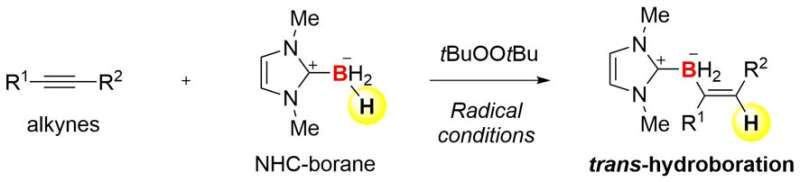
Borane (BH3) and its derivatives stabilized by ligands are the simplest boron compounds. They can react with a carbon-carbon triple bond (alkyne, C-C) to yield an insertion product (H-C=C-BH2). This reaction, called hydroboration, is a powerful method to prepare organoboron compounds. However, the reaction typically provides only a cis-product, which means that H and BH2 are introduced on the same side of the triple bond. That is, it is difficult to synthesize a trans-hydroboration product with the opposite geometric configuration. Previously, there have been very limited examples of trans-hydroboration of alkynes.
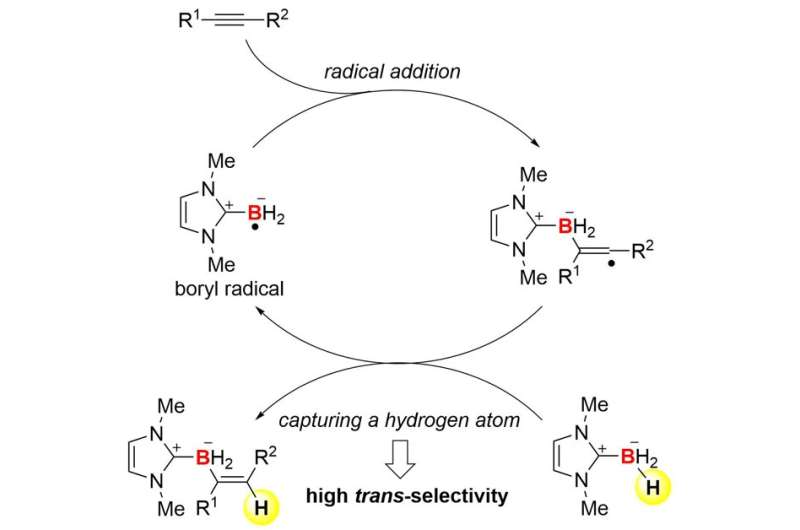
The collaboration team between Kanazawa University and the University of Pittsburgh succeeded in the development of trans-hydroboration reactions of alkynes based on radical chemistry (Figure 1). The team employed N-heterocyclic carbene boranes (NHC-boranes) to combine hydroboration with radical chemistry. NHC-boranes can be handled easily because of their stability, and they are good precursors of boryl radicals (boron-centered radicals). Indeed, an NHC-boryl radical can be readily formed by simple thermolysis in the presence of inexpensive commercial di-tert-butyl peroxide. The radical can add to an alkyne to form a carbon-boron (C-B) bond and a new carbon radical (Figure 2). Trans-selectivity in hydroboration is kinetically induced when the highly reactive carbon radical captures a hydrogen atom from the starting NHC-borane. As a result, the overall process establishes a radical chain cycle. This mechanism is quite different from that of known hydroboration reactions.
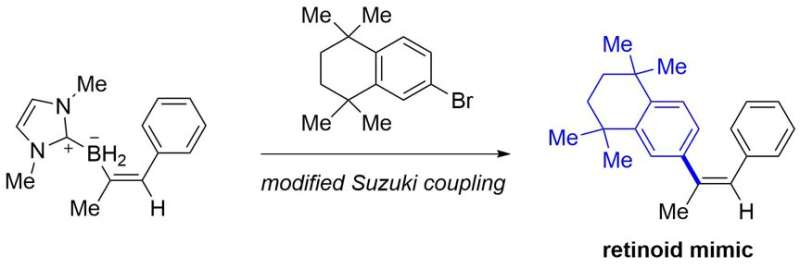
From easily available alkynes, the present protocol provides various bench-stable alkenyl borane compounds that are not easily accessible by known methods. Some of them can be converted to retinoid mimics, which are drug candidates, by modified Suzuki coupling (Figure 3).
Organoboron compounds synthesized by the present method will give access to new boron-containing π-systems by further chemical transformation. Therefore, this type of trans-hydroboration reaction will facilitate advances in medicinal chemistry and materials science. From the standpoint of pure chemistry, this study expands the potential of radicals in synthetic chemistry. In short, we illustrate that radicals are capable of controlling chemical reactions precisely despite their extreme reactivity.
Dennis Curran (University of Pittsburgh), a collaborator in this study, says, "It has been a pleasure to be involved in this collaboration, which was led by the Kanazawa team. The new reaction that we have discovered is unique, and I am excited about its prospects for extension in the directions of both basic research and practical applications."
More information: Masaki Shimoi et al, Radical trans -Hydroboration of Alkynes with N-Heterocyclic Carbene Boranes Angewandte Chemie International Edition (2018). DOI: 10.1002/anie.201804515
Journal information: Angewandte Chemie International Edition
Provided by Kanazawa University